Nanoplate vs. Droplet Digital PCR: A 2025 Comparison of Precision, Accuracy, and Application
This article provides a comprehensive analysis of two principal digital PCR technologies: nanoplate-based and droplet-based dPCR.
Nanoplate vs. Droplet Digital PCR: A 2025 Comparison of Precision, Accuracy, and Application
Abstract
This article provides a comprehensive analysis of two principal digital PCR technologies: nanoplate-based and droplet-based dPCR. Tailored for researchers and drug development professionals, it synthesizes recent 2025 data to compare the precision, accuracy, limitations, and optimal applications of each platform. We cover foundational principles, methodological workflows across diverse fields like oncology, infectious disease, and GMO detection, and offer troubleshooting guidance. The review concludes with a validated comparative outlook to inform platform selection for both research and quality-controlled environments, supporting robust experimental design and molecular diagnostics.
The Technology Divide: Understanding Nanoplate and Droplet dPCR Core Principles
The polymerase chain reaction (PCR) has undergone a remarkable evolution since its invention, transforming from a qualitative tool to a precision quantification platform. Quantitative PCR (qPCR) revolutionized molecular biology by enabling relative quantification of nucleic acids, but its dependence on standard curves introduced variability. The emergence of digital PCR (dPCR) represents the third generation of PCR technology, offering absolute quantification without external references by applying Poisson statistics to thousands of parallel reactions [1] [2]. This advancement is particularly valuable for applications requiring high precision, including liquid biopsy analysis, pathogen detection, and copy number variation studies [3] [4]. Two principal dPCR platforms have emerged: droplet digital PCR (ddPCR) and nanoplate-based dPCR, each with distinct technological approaches and performance characteristics that researchers must consider for their specific applications [5] [6].
Technical Comparison: Nanoplate-Based vs. Droplet Digital PCR
Fundamental Partitioning Mechanisms
The core distinction between nanoplate and droplet dPCR platforms lies in their partitioning methodologies:
Nanoplate-based dPCR utilizes microfluidic chips containing fixed nanowells (typically 8,500-26,000 partitions) that evenly distribute the reaction mixture in equal volumes within a closed system [6] [7]. This approach minimizes contamination risk and simplifies workflow.
Droplet digital PCR (ddPCR) generates thousands to millions of nanoliter-sized water-in-oil droplets (10-100 pL each) that function as individual reaction chambers [2] [6]. While offering high partition numbers, droplets face challenges with size variability, coalescence, and shearing during thermal cycling [6].
Workflow and Operational Considerations
The practical implementation of these technologies reveals significant operational differences:
Table 1: Workflow Comparison Between dPCR Platforms
| Parameter | Nanoplate-based dPCR | Droplet Digital PCR (ddPCR) |
|---|---|---|
| Workflow Steps | Single-step process similar to qPCR | Multiple instrument transfers required |
| Partitioning | Automated in integrated instrument | Separate droplet generator needed |
| Amplification & Reading | Integrated thermocycling and imaging | Requires transfer to separate thermocycler and reader |
| Hands-on Time | Minimal | More extensive |
| Risk of Contamination | Lower due to closed system | Higher due to multiple transfer steps |
| Throughput | High (up to 96 samples per run) | Variable (typically lower) |
| Ease of Use | More accessible, minimal training | Requires specialized pipetting skills |
Performance Analysis: Precision and Sensitivity in Research Applications
Analytical Performance Metrics
Direct comparisons of nanoplate-based (QIAcuity) and droplet-based (QX200) dPCR platforms reveal nuanced performance characteristics:
Table 2: Analytical Performance Comparison
| Performance Metric | Nanoplate-based dPCR | Droplet Digital PCR | Experimental Context |
|---|---|---|---|
| Limit of Detection (LOD) | ~0.39 copies/μL [8] | ~0.17 copies/μL [8] | Synthetic oligonucleotides |
| Limit of Quantification (LOQ) | 1.35 copies/μL [8] | 4.26 copies/μL [8] | Synthetic oligonucleotides |
| Specificity | 99.62% [5] | 100% [5] | CDH13 methylation detection |
| Sensitivity | 99.08% [5] | 98.03% [5] | CDH13 methylation detection |
| Correlation Between Platforms | r = 0.954 [5] | r = 0.954 [5] | Methylation level correlation |
| Precision (CV with EcoRI) | 0.6-27.7% [8] | 2.5-62.1% [8] | Paramecium tetraurelia DNA |
| Precision (CV with HaeIII) | 1.6-14.6% [8] | <5% [8] | Paramecium tetraurelia DNA |
Reproducibility in Clinical Applications
Studies comparing both platforms for clinical diagnostics demonstrate their reliability:
Liquid Biopsy Analysis: A 2023 study comparing dPCR platforms for detecting EGFR and RAS mutations in liquid biopsy samples from lung and colorectal cancer patients found moderate agreement between ddPCR and nanoplate dPCR. However, solid-phase nanoplate dPCR demonstrated higher sensitivity in detecting mutated cases (100% for EGFR mutations vs. 58.8% for ddPCR) [4].
Respiratory Virus Detection: During the 2023-2024 "tripledemic," dPCR showed superior accuracy to real-time RT-PCR, particularly for high viral loads of influenza A, influenza B, and SARS-CoV-2, with nanoplate-based systems offering greater automation and throughput [3] [9].
Experimental Protocols: Methodologies for Platform Comparison
DNA Methylation Analysis Protocol
Research comparing dPCR platforms for methylation-specific detection typically follows this methodology [5]:
- Sample Preparation: DNA is extracted from patient samples (e.g., 141 FFPE breast cancer tissues) using commercial kits (DNeasy Blood and Tissue Kit).
- Bisulfite Conversion: DNA treatment with bisulfite reagents (EpiTect Bisulfite Kit) converts unmethylated cytosines to uracils while preserving methylated cytosines.
- Assay Design: Primers and probes target specific CpG sites in genes of interest (e.g., CDH13 promoter region), with separate probes for methylated (FAM-labeled) and unmethylated (HEX-labeled) sequences.
- dPCR Setup:
- Nanoplate-based: 12μL reactions with 4× Probe PCR Master Mix pipetted into 24-well nanoplate (8,500 partitions/well)
- Droplet-based: 20μL reactions with Supermix for Probes partitioned into ~20,000 droplets
- Thermocycling: Initial activation (95°C for 2min), 40 cycles of denaturation (95°C for 15s), and combined annealing/extension (57°C for 1min)
- Analysis: Threshold determination and calculation of methylation percentage as ratio of FAM-positive to total positive partitions
Workflow Visualization
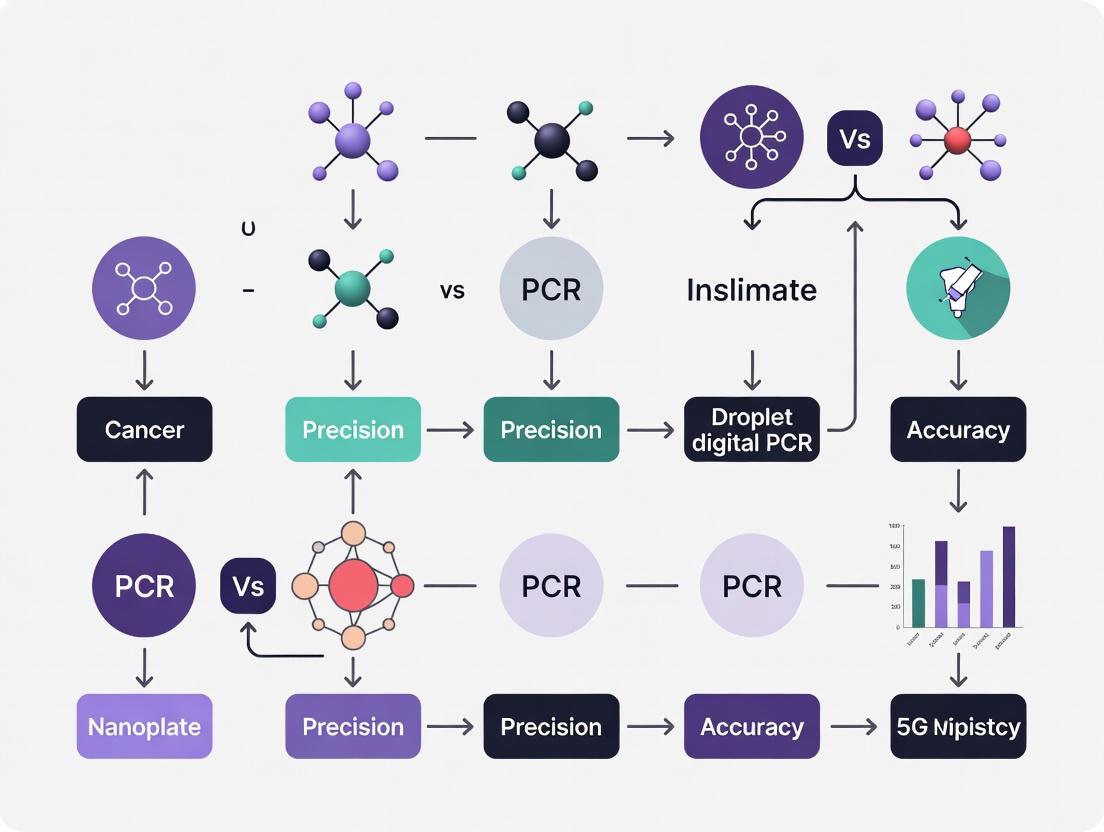
The following diagram illustrates the fundamental procedural differences between the two dPCR platforms and their predecessor, qPCR:
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for dPCR Experiments
| Reagent/Material | Function | Example Products |
|---|---|---|
| DNA Extraction Kits | Nucleic acid purification from various sample types | DNeasy Blood & Tissue Kit, MagMax Viral/Pathogen Kit [5] [3] |
| Bisulfite Conversion Kits | DNA modification for methylation analysis | EpiTect Bisulfite Kit [5] |
| dPCR Master Mixes | Optimized buffers, enzymes, and nucleotides for partition PCR | QIAcuity 4× Probe PCR Master Mix, Supermix for Probes [5] |
| Fluorescent Probes | Sequence-specific detection with fluorophore-quencher pairs | FAM-labeled M-probes, HEX-labeled UnM-probes [5] |
| Primer Sets | Target-specific amplification | Custom-designed primers for genes of interest [5] [7] |
| Reference Materials | Assay validation and standardization | Synthetic oligonucleotides, standardized DNA [8] [7] |
The evolution from qPCR to dPCR represents a significant advancement in molecular quantification, offering researchers unprecedented precision and absolute quantification capabilities. Both nanoplate-based and droplet digital PCR platforms demonstrate excellent analytical performance with strong correlation in quantitative measurements [5] [8]. The choice between platforms depends on specific research requirements:
Nanoplate-based dPCR offers advantages in workflow simplicity, reduced contamination risk, and higher throughput, making it ideal for clinical diagnostics and high-volume applications [6] [4].
Droplet digital PCR provides extremely high partition numbers and established protocols, potentially offering superior sensitivity for ultra-rare target detection, though with more complex workflow requirements [8] [6].
For most laboratory settings, particularly those transitioning from qPCR, nanoplate-based systems provide a more accessible entry to digital PCR technology with comparable performance metrics and streamlined operation. As both technologies continue to evolve, their complementary strengths will further expand the applications of absolute nucleic acid quantification in research and clinical diagnostics.
Droplet Digital PCR (ddPCR) represents a significant advancement in nucleic acid quantification by partitioning samples into thousands of nanoliter-scale water-in-oil emulsion droplets. This guide examines the core mechanics of ddPCR partitioning and objectively compares its performance against nanoplate-based dPCR systems, with a specific focus on precision and accuracy metrics relevant to research and drug development applications. Experimental data from recent studies demonstrate that while both platforms offer high sensitivity and reproducibility, key differences in their partitioning mechanics and operational parameters significantly influence their performance across various applications.
Droplet Digital PCR (ddPCR) is a digital PCR method utilizing a water-oil emulsion droplet system. The fundamental principle involves dispersing a PCR reaction mixture containing template DNA into tens of thousands of nanoliter-sized droplets, creating independent reaction chambers where amplification occurs. Each droplet functions as an individual test tube, with the massive sample partitioning enabling absolute quantification of target nucleic acid molecules without requiring standard curves. This water-in-oil emulsion system is typically generated by microfluidic technologies or interface vertical vibration methods that create uniform droplets in a water-oil emulsion to form the partitions that separate template DNA molecules [10] [11].
The partitioning process is facilitated by specialized oil phase compositions containing specific surfactants that stabilize the emulsion. Optimal formulations include: (1) macromolecular non-ionic surfactants with low hydrophilic-lipophilic balance (HLB) values and polydimethylsiloxane backbones (e.g., ABIL EM 90); (2) polyethylene glycol small molecule non-ionic surfactants with high HLB values (e.g., Tween series); (3) anti-evaporation agents (e.g., polyisobutenol with molecular weight of 1300); and (4) mineral oil as a base oil [11]. This composition ensures droplets have uniform size, excellent thermal stability, no evaporation or fusion during thermal cycling, and no inhibition of PCR amplification.
Following endpoint PCR amplification, droplets are analyzed individually using flow cytometry principles. droplets containing the target nucleic acid sequence generate fluorescent signals, while those without the target remain non-fluorescent. The proportion of positive droplets enables absolute quantification of the target molecule using Poisson statistics, providing sensitivity down to single nucleic acid molecules [10] [12].
Comparative Performance Analysis: ddPCR vs. NanoplatedPCR
Sensitivity and Detection Limits
Table 1: Comparison of Detection and Quantification Limits Between dPCR Platforms
| Parameter | QX200 ddPCR (Bio-Rad) | QIAcuity One ndPCR (QIAGEN) | Experimental Context |
|---|---|---|---|
| Limit of Detection (LOD) | 0.17 copies/μL input (3.31 copies/reaction) | 0.39 copies/μL input (15.60 copies/reaction) | Synthetic oligonucleotides with 40μL (ndPCR) and 20μL (ddPCR) reactions [8] |
| Limit of Quantification (LOQ) | 4.26 copies/μL input (85.2 copies/reaction) | 1.35 copies/μL input (54 copies/reaction) | Based on 3rd degree polynomial model fit [8] |
| Dynamic Range | 0.17->3000 copies/μL input | 0.39->3000 copies/μL input | Six dilution levels from synthetic oligonucleotides [8] |
| ctDNA Detection Rate | 58.5% (24/41 patients) | 36.6% (15/41 patients) | Localized rectal cancer baseline plasma (p=0.00075) [13] [14] |
Precision and Accuracy Metrics
Table 2: Precision and Accuracy Comparisons Across Platforms
| Performance Measure | QX200 ddPCR | QIAcuity One ndPCR | Notes |
|---|---|---|---|
| Accuracy (R²adj vs. expected copies) | 0.99 | 0.98 | Synthetic oligonucleotide dilution series [8] |
| Precision (CV range) | 6-13% | 7-11% | Across dilution levels above LOQ [8] |
| Precision with EcoRI restriction enzyme | CV: 2.5-62.1% | CV: 0.6-27.7% | Paramecium tetraurelia DNA, varying cell numbers [8] |
| Precision with HaeIII restriction enzyme | CV: <5% | CV: 1.6-14.6% | Significant improvement for ddPCR with alternative enzyme [8] |
| Operational Cost Factor | 5-8.5x lower than NGS | Higher cost than ddPCR | Relative cost comparison for ctDNA detection [13] |
The data reveal platform-specific strengths, with ddPCR demonstrating superior detection capabilities for circulating tumor DNA in clinical applications, while ndPCR showed marginally better quantification limits in controlled conditions. Restriction enzyme selection significantly impacted ddPCR precision, with HaeIII dramatically improving performance over EcoRI, while ndPCR showed less enzyme dependency [8].
Figure 1: Comparative Workflow of ddPCR and Nanoplate dPCR Platforms. Both technologies follow similar initial and final steps but diverge in their partitioning mechanisms, which significantly influences their performance characteristics and application suitability.
Experimental Protocols and Methodologies
ddPCR Partitioning and Emulsion Formation
The core mechanical process of ddPCR partitioning involves creating a stable water-in-oil emulsion. The aqueous phase contains template DNA, forward and reverse primers, double-stranded DNA fluorescent dye or TaqMan fluorescent probes, DNA polymerase, and PCR reaction buffer. This aqueous solution is mixed with the oil phase composition in a specific ratio [11].
For the QX200 system (Bio-Rad), each 20μL reaction mixture is partitioned into approximately 20,000 nanoliter-sized droplets through a microfluidic droplet generator. The oil phase composition is critical for maintaining droplet integrity during thermal cycling. Optimal formulations include 2-5% w/w macromolecular nonionic surfactant with polydimethylsiloxane backbone (e.g., ABIL EM 90), 0.05-0.2% w/w polyethylene glycol small molecule nonionic surfactant (e.g., Tween series), 1-5% w/w anti-evaporation agent (e.g., polyisobutylene PB1300), with the balance being mineral oil [11]. This formulation ensures uniform droplet size, prevents droplet evaporation and fusion, and doesn't inhibit PCR amplification.
After droplet generation, endpoint PCR amplification is performed using conventional thermal cycling conditions: 95°C pre-denaturation for 5 minutes, followed by 40 cycles of 95°C denaturation for 20 seconds and 55°C annealing/extension for 40 seconds [11].
ctDNA Detection Protocol (Rectal Cancer Study)
In a recent comparative study analyzing ctDNA detection in non-metastatic rectal cancer, researchers implemented the following protocol [13] [14]:
Sample Collection: Pre-therapy plasma samples were collected from patients using 3 × 9 mL of blood drawn into Streck Cell Free DNA BCT vacuum tubes before any neoadjuvant therapy.
cfDNA Extraction: Cell-free DNA was extracted from plasma samples using standard silica membrane-based methods.
Tumor Mutation Identification: Tumor tissue samples underwent sequencing using the Ion AmpliSeq Cancer Hotspot Panel v2 (HS1) to identify patient-specific mutations.
ddPCR Assay Design: Custom ddPCR assays were designed targeting one to two mutations with the highest variant allele frequencies identified in matched primary tumor NGS analysis.
ddPCR Setup: Reactions were prepared using 2-9μL of extracted DNA partitioned into 20,000 droplets. Absolute quantification of targeted cfDNA was calculated based on PCR-positive and PCR-negative droplets using Poisson statistics.
NGS Comparison: The same HS1 panel sequencing was performed for ctDNA detection, with variant calling threshold lowered to 0.01% variant allele frequency (VAF) based on ddPCR results.
This tumor-informed ddPCR approach demonstrated significantly higher detection rates (58.5%) compared to the tumor-uninformed NGS panel (36.6%) in baseline plasma samples from the development cohort (p=0.00075) [13].
Cross-Platform Precision Assessment
A 2025 study directly compared the precision of QX200 ddPCR and QIAcuity One ndPCR using both synthetic oligonucleotides and DNA extracted from varying cell numbers of Paramecium tetraurelia [8]:
DNA Material Preparation: Synthetic oligonucleotides were diluted across 11 concentration levels from <0.5 copies/μL to >3000 copies/μL input. Paramecium tetraurelia DNA was extracted from precisely counted cell numbers.
Restriction Enzyme Evaluation: Two restriction enzymes (EcoRI and HaeIII) were tested for their impact on gene copy number quantification, particularly for accessing tandemly repeated genes.
Parallel Amplification: Both platforms performed amplifications using identical primer sets and reaction conditions where possible, with 20μL reactions for ddPCR and 40μL reactions for ndPCR.
Data Analysis: Limit of detection (LOD) and limit of quantification (LOQ) were determined for both platforms. Precision was assessed through coefficient of variation (CV) calculations across replicate measurements.
This rigorous methodological approach enabled direct comparison of platform performance using the same DNA material, revealing important practical considerations for researchers selecting appropriate digital PCR systems for specific applications.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for ddPCR Experimental Workflows
| Reagent/Chemical | Function in ddPCR Workflow | Example Products/Formulations |
|---|---|---|
| Oil Phase Composition | Creates stable water-in-oil emulsion for partitioning | Mineral oil base with 2-5% ABIL EM 90, 0.05-0.2% Tween surfactant, 1-5% polyisobutylene PB1300 [11] |
| Surface-Active Agents | Stabilize emulsion droplets during thermal cycling | Macromolecular non-ionic surfactants with polydimethylsiloxane backbone (low HLB); PEG small molecule surfactants (high HLB) [11] |
| Anti-Evaporation Agents | Prevent droplet evaporation during high-temperature steps | Polyisobutenol with molecular weight of 1300 (e.g., polyisobutylene H300, PB1300) [11] |
| Restriction Enzymes | Enhance target accessibility for quantification | HaeIII demonstrated superior precision vs. EcoRI in ddPCR applications [8] |
| Fluorescent Probes/Dyes | Enable endpoint detection of amplified targets | TaqMan hydrolysis probes, EvaGreen dsDNA binding dyes [8] |
| Reference Standard Materials | Validate assay performance and quantification accuracy | Characterized cell lines with known mutation status (e.g., EGFR T790M) [10] [12] |
Figure 2: ddPCR Reagent System and Partitioning Mechanism. The interplay between oil phase components and aqueous phase reagents creates stable emulsion droplets that serve as individual reaction chambers, enabling absolute quantification of target nucleic acids.
The core mechanics of ddPCR's water-in-oil emulsion partitioning provide distinct advantages for applications requiring absolute quantification of rare targets and subtle genetic variations. Recent comparative studies demonstrate that ddPCR offers superior detection sensitivity for clinical applications like ctDNA analysis compared to both NGS and nanoplate dPCR systems. However, nanoplate dPCR shows marginally better quantification limits in some controlled conditions. The precision of both platforms is significantly influenced by methodological factors, particularly restriction enzyme selection in ddPCR applications. These performance comparisons provide researchers and drug development professionals with critical data for selecting appropriate digital PCR methodologies based on specific application requirements, whether prioritizing ultimate detection sensitivity, quantification precision, or operational considerations.
Digital PCR (dPCR) represents the third generation of PCR technology, enabling absolute quantification of nucleic acids without the need for standard curves. The core principle relies on partitioning a PCR reaction into thousands of individual reactions, allowing single-molecule detection via Poisson statistics [15]. Two primary partitioning methods have emerged: water-in-oil droplet emulsification (ddPCR) and fixed microchamber arrays (nanoplate dPCR) [15]. Droplet-based systems, such as the Bio-Rad QX200, generate approximately 20,000 nanoliter-sized droplets through water-oil emulsion [16]. In contrast, nanoplate-based systems, exemplified by the QIAGEN QIAcuity, utilize microfluidic chips containing fixed arrays of nanoscale chambers—typically 8,500 or 26,000 partitions per well [17]. This fundamental difference in partitioning mechanics creates distinct operational and performance characteristics that researchers must consider when selecting platforms for precision applications in drug development and clinical diagnostics.
Technical Mechanics of Nanoplate Digital PCR
Microfluidic Chamber Array Architecture
The nanoplate digital PCR system employs a sophisticated microfluidic architecture where partitions are physically etched into solid chips. These fixed microchambers are arranged in precise arrays, with the QIAcuity system offering nanoplates in two primary configurations: 24-well plates with approximately 8,500 partitions per well and 96-well plates with approximately 26,000 partitions per well [18] [17]. This fixed array design ensures consistent partition volume and geometry across all samples, eliminating the variability inherent in droplet generation processes. The partitions are permanently sealed during the amplification process, preventing well-to-well contamination and partition merging that can occur with emulsion-based systems [17]. The entire process—from partitioning and thermocycling to imaging—occurs within an integrated instrument system, creating a streamlined "sample-in, results-out" workflow [16] that significantly reduces hands-on time compared to droplet-based methods.
Comparative Workflow Efficiency
The integrated nature of nanoplate dPCR systems provides significant operational advantages over droplet-based platforms, particularly in regulated environments such as quality control laboratories. The workflow comparison reveals substantial differences in time requirements and manual intervention. Where droplet digital PCR typically requires multiple instruments (droplet generator, thermal cycler, and droplet reader) and a total process time of 6-8 hours, nanoplate systems complete the entire process in under 90 minutes within a single integrated instrument [16]. This streamlined workflow reduces potential contamination risks through fewer manual transfer steps and decreases the technical expertise required for operation. The fixed nanoplate format closely resembles traditional qPCR plates, making the transition to digital PCR more accessible for laboratories with existing qPCR experience [19].
Diagram 1: Workflow comparison between droplet and nanoplate digital PCR platforms.
Experimental Performance Comparison
Sensitivity and Precision Metrics
Multiple independent studies have directly compared the analytical performance of nanoplate and droplet digital PCR systems across various applications. In a comprehensive comparison using synthetic oligonucleotides and DNA from Paramecium tetraurelia, both platforms demonstrated similar detection capabilities, with the QIAcuity nanoplate system showing a limit of detection (LOD) of 0.39 copies/μL compared to 0.17 copies/μL for the QX200 droplet system [8]. For limit of quantification (LOQ), the nanoplate system demonstrated 1.35 copies/μL versus 4.26 copies/μL for the droplet system [8]. Precision measurements using coefficient of variation (CV) showed that both platforms produced highly reproducible results, with CVs ranging between 6-13% for ddPCR and 7-11% for ndPCR across most concentration levels above the LOQ [8]. Notably, the nanoplate system maintained more consistent precision across a wider concentration range, while the droplet system achieved optimal precision at mid-range concentrations around 270 copies/μL [8].
Table 1: Performance comparison of nanoplate vs. droplet digital PCR systems
| Performance Parameter | Nanoplate dPCR (QIAcuity) | Droplet ddPCR (QX200) | Experimental Context |
|---|---|---|---|
| Limit of Detection | 0.39 copies/μL | 0.17 copies/μL | Synthetic oligonucleotides [8] |
| Limit of Quantification | 1.35 copies/μL | 4.26 copies/μL | Synthetic oligonucleotides [8] |
| Precision (CV Range) | 7-11% | 6-13% | Multiple dilution levels [8] |
| Restriction Enzyme Impact | Minimal CV difference | Significant improvement with HaeIII | Paramecium tetraurelia DNA [8] |
| Correlation with ddPCR | r = 0.954 | Reference | CDH13 methylation analysis [5] |
| Partition Number | 8,500-26,000 | ~20,000 | Platform specifications [16] [17] |
Method-Specific Performance Applications
GMO Quantification in Food Analysis
In food authentication applications, both dPCR platforms demonstrated excellent performance for quantifying genetically modified organisms. An in-house validation of duplex methods for detecting MON-04032-6 and MON89788 soybean events showed that all evaluated data and validation parameters agreed with acceptance criteria according to JRC Guidance documents on both platforms [20]. The nanoplate system successfully transferred optimized assays from droplet-based systems with equivalent performance, demonstrating particular strengths in multiplexing efficiency and streamlined workflow for routine testing [20].
DNA Methylation Analysis
In epigenetics research, a comparison of the two platforms for CDH13 gene methylation analysis in 141 breast cancer tissue samples revealed a strong correlation (r = 0.954) between methylation levels measured by both methods [5]. The QIAcuity nanoplate system demonstrated 99.62% specificity and 99.08% sensitivity, while the QX200 droplet system reached 100% specificity and 98.03% sensitivity [5]. Researchers concluded that while both methods yielded comparable, highly sensitive experimental data, the selection criteria for an optimal platform should consider factors such as workflow time and complexity, instrument requirements, and reanalysis options [5].
Residual Cell Detection in Cell Therapy
For detecting trace residual cells in cell therapy manufacturing, both dPCR platforms exhibited superior linearity and markedly lower coefficients of variation compared to qPCR [21]. While minor differences in absolute copy number were observed, both dPCR systems demonstrated comparable analytical performance for detecting undifferentiated induced pluripotent stem cells (iPSCs) and virus-producing cells at dilution levels as low as 1:10^6 [21]. This application highlights the critical importance of precise quantification in advanced therapy medicinal products (ATMPs), where both digital PCR technologies enable robust detection at trace levels.
Table 2: Application-specific performance comparison
| Application Area | Nanoplate dPCR Advantages | Droplet ddPCR Advantages | Study Details |
|---|---|---|---|
| Food Allergen Detection | Sensitivity improved by one order of magnitude vs. qPCR; Not affected by food processing | - | Sesame detection in dough/biscuits [22] |
| Meat Authentication | LOD of 0.1% (w/w); More sensitive than 1% LOD of qPCR; Better resistance to inhibitors | - | Pork and chicken in processed meats [19] |
| Periodontal Pathogen Detection | Lower intra-assay variability (median CV: 4.5%) than qPCR; Effective multiplexing | - | Three periodontal pathogens [18] |
| Cell Therapy Safety | Comparable performance to ddPCR for residual iPSC detection | Extensive validation history in regulatory submissions | CAR-T and iPSC-based therapies [16] [21] |
Research Reagent Solutions for Nanoplate dPCR
Successful implementation of nanoplate digital PCR requires specific reagent systems optimized for the fixed-partition architecture. Key components include:
- QIAcuity Nanoplate 26k: Microfluidic plates with 24 or 96 wells providing approximately 26,000 partitions per well; fixed partitions prevent well-to-well contamination and variability [18] [17].
- QIAcuity Probe PCR Kit: Master mix specifically formulated for nanoplate partitioning, containing DNA polymerase, dNTPs, and optimized buffers for efficient amplification in nanoscale chambers [18].
- Restriction Enzymes (e.g., HaeIII): Critical for reducing fragment size and improving access to tandemly repeated genes; shown to significantly improve precision, especially in complex samples [8].
- Anza 52 PvuII Restriction Enzyme: Used in multiplex pathogen detection to enhance target accessibility; particularly important for complex genomic targets [18].
- Volume Precision Factor v9.0: Software-based correction factor applied according to manufacturer's instructions to improve accuracy of concentration calculations by accounting for partition volume variations [18].
The core mechanical difference between nanoplate and droplet digital PCR—fixed microchamber arrays versus generated emulsion droplets—translates into distinct operational and performance characteristics. Current comparative studies indicate that both technologies deliver comparable sensitivity, precision, and accuracy for most molecular applications [8] [5] [20]. The nanoplate system offers significant advantages in workflow integration, reduced hands-on time, and minimized contamination risk through its fixed-partition, all-in-one instrument design [16] [17]. Conversely, droplet systems benefit from extensive validation history and established regulatory submission precedents [16]. For researchers prioritizing streamlined operations in quality control environments or requiring efficient multiplexing, nanoplate dPCR presents a compelling solution. For applications demanding the highest possible sensitivity or building upon extensively documented droplet-based protocols, droplet digital PCR remains a robust choice. The decision ultimately hinges on specific application requirements, regulatory considerations, and operational constraints within the research or quality control environment.
Digital PCR (dPCR) has emerged as a powerful tool for absolute nucleic acid quantification, offering superior precision and accuracy compared to traditional quantitative PCR. The core principle of dPCR involves partitioning a sample into thousands of individual reactions, enabling binary detection of target molecules through endpoint amplification. This partitioning step is fundamental, and the two predominant methodologies—dynamic droplets and static nanowells—define the operational characteristics, performance metrics, and practical applications of different dPCR platforms. Dynamic droplet systems, such as droplet digital PCR (ddPCR), generate nanoliter-sized water-in-oil emulsions, while static nanowell systems, like the QIAcuity, employ microfluidic chips with fixed physical compartments. This guide provides an objective comparison of these partitioning technologies, contextualized within the broader thesis of achieving precision and accuracy in nanoplate versus droplet digital PCR research, to inform scientists and drug development professionals in their platform selection process.
Technical Comparison of Partitioning Technologies
The fundamental difference between droplet and nanowell dPCR lies in the mechanism of partition creation and management. The following table summarizes the core technical characteristics of each partitioning method.
Table 1: Technical Specifications of Dynamic Droplet vs. Static Nanowell dPCR
| Feature | Dynamic Droplets (ddPCR) | Static Nanowells (e.g., QIAcuity) |
|---|---|---|
| Partition Type | Transient, emulsion-based droplets [3] | Fixed, physical nanowells on a chip [3] |
| Partition Volume | Nanoliter-sized [23] | Nanoliter-sized (e.g., 1 nL) [23] |
| Typical Partition Count | Up to 20,000 droplets per sample [13] | ~26,000 nanowells per sample [3] |
| Core Workflow | Microfluidic droplet generation, end-point PCR, sequential droplet reading [13] | Microfluidic chip loading, end-point PCR, simultaneous imaging of all nanowells [3] [23] |
| Liquid Handling | Requires droplet generation and transfer steps [13] | Integrated microfluidic cartridge for simplified loading [3] |
| Throughput & Automation | Lower throughput; reduced automation [3] | Higher throughput; amenable to automated workflows [3] |
Performance and Application in Biomedical Research
The technical distinctions between these platforms translate directly into performance differences that impact their utility in specific research and diagnostic applications. Both technologies have been rigorously evaluated in recent studies.
Quantitative Precision and Diagnostic Performance
A 2025 study directly compared dPCR and real-time RT-PCR for detecting respiratory viruses during the 2023–2024 tripledemic. The research, which utilized a nanowell-based system (QIAcuity), found that dPCR demonstrated superior accuracy, particularly for high viral loads of influenza A, influenza B, and SARS-CoV-2, and for medium loads of RSV. It also showed greater consistency and precision than Real-Time RT-PCR, especially in quantifying intermediate viral levels [3]. This enhanced precision is a direct benefit of the high number of partitions, which allows for more robust statistical analysis of target molecule concentration.
Another 2025 study on circulating tumor DNA (ctDNA) in rectal cancer highlighted the performance of droplet-based ddPCR. The study reported that ddPCR detected ctDNA in 58.5% (24/41) of baseline plasma samples, a rate significantly higher (p = 0.00075) than the 36.6% (15/41) detection rate achieved by a next-generation sequencing (NGS) panel. This demonstrates the high sensitivity of ddPCR for detecting low-abundance targets in complex biological samples like plasma [13].
Application in Complex Sample Analysis and Antimicrobial Testing
The nanoarray platform, a form of static nanowell dPCR, has been successfully combined with high-resolution melt (HRM) analysis and machine learning for broad bacterial identification and antimicrobial susceptibility testing (AST). This dPCR-HRM workflow allows for the analysis of polymicrobial samples by ensuring that each melt curve is generated from a single bacterial species within an individual nanowell. The quantitative precision of this nanoarray platform enables rapid phenotypic AST after only 30 minutes of antibiotic exposure, a significant acceleration compared to culture-based methods [23].
Table 2: Experimental Performance Benchmarks in Recent Studies
| Application | Platform Type | Key Performance Metric | Result | Source |
|---|---|---|---|---|
| Respiratory Virus Detection | Static Nanowells | Superior accuracy for high viral loads | Demonstrated for Influenza A, B, & SARS-CoV-2 | [3] |
| ctDNA Detection in Rectal Cancer | Dynamic Droplets | Detection rate in baseline plasma | 58.5% (ddPCR) vs. 36.6% (NGS) | [13] |
| Antimicrobial Susceptibility Testing | Static Nanowells (Nanoarray) | Required antibiotic exposure time | ~30 minutes | [23] |
| Polymerase Inhibition Resistance | Digital PCR (General) | Robustness in complex samples | More resistant to inhibitors than qPCR | [24] |
Experimental Protocols for Platform Evaluation
To ensure the reliability and comparability of data generated by these platforms, standardized experimental protocols are critical. The following methodologies are adapted from recent publications to serve as a guide for benchmarking studies.
Protocol 1: Comparative Quantification of Viral Load
This protocol is derived from a study comparing dPCR and RT-PCR for respiratory virus quantification [3].
- Sample Preparation: Collect respiratory samples (e.g., nasopharyngeal swabs) and stratify them based on RT-PCR cycle threshold (Ct) values into high (Ct ≤ 25), medium (Ct 25.1–30), and low (Ct > 30) viral load categories.
- Nucleic Acid Extraction: Perform RNA extraction using an automated platform (e.g., KingFisher Flex system) with a viral/pathogen nucleic acid isolation kit.
- dPCR Setup (Nanowell System):
- Use a integrated dPCR system (e.g., QIAcuity).
- Prepare a PCR mixture using a commercially available multiplex assay targeting the viruses of interest (e.g., Influenza A, Influenza B, RSV, SARS-CoV-2).
- Load the mixture into a nanowell plate (e.g., generating ~26,000 partitions) and run the endpoint PCR protocol as per the manufacturer's instructions.
- Data Analysis: Use the instrument's suite software (e.g., QIAcuity Suite Software) to calculate the absolute copy number per microliter of each target based on the count of positive and negative partitions.
Protocol 2: Sensitivity Detection for Low-Abundance Targets
This protocol is based on studies detecting ctDNA and viral genomes, showcasing the sensitivity of droplet-based dPCR [13] [24].
- Sample Preparation: Collect plasma samples in cell-free DNA blood collection tubes. Isociate cell-free DNA (cfDNA) from plasma using a commercial kit.
- Assay Design (Tumor-Informed): For ctDNA, sequence the primary tumor tissue using a next-generation sequencing (NGS) cancer hotspot panel. Identify the top one or two somatic mutations with the highest variant allele frequency for which ddPCR probes can be designed.
- ddPCR Setup:
- Prepare a reaction mix containing the extracted cfDNA, ddPCR supermix, and mutation-specific, fluorescently labeled probes.
- Generate droplets (typically ~20,000 droplets per sample) using an automated droplet generator.
- Transfer the emulsified sample to a 96-well plate and perform endpoint PCR amplification.
- Droplet Reading and Analysis: Place the plate in a droplet reader, which counts the number of PCR-positive and PCR-negative droplets for each fluorescent channel. The absolute concentration of the mutant DNA fragment (copies/μL) is calculated using Poisson statistics.
Workflow and Signaling Pathway Visualization
The core differentiator between the two dPCR methodologies is the workflow, from sample partitioning to result analysis. The following diagram illustrates the distinct steps involved in each pathway.
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of either dPCR technology requires a set of core reagents and materials. The following table details these essential components and their functions.
Table 3: Key Research Reagent Solutions for Digital PCR
| Item | Function | Application Notes |
|---|---|---|
| Digital PCR Supermix | Provides optimized buffer, enzymes, and dNTPs for efficient amplification in partitioned reactions. | Must be compatible with the specific platform (droplet or nanowell) and detection chemistry (probe-based or EvaGreen) [3] [13]. |
| Primer & Probe Assays | Target-specific oligonucleotides for nucleic acid amplification and detection. | Can be uniplex or multiplex; hydrolysis (TaqMan) probes are commonly used. Mutation-specific probes are essential for ctDNA detection [13] [24]. |
| Microfluidic Cartridges/Chips | The physical substrate that creates the static nanowells for partitioning. | Specific to the instrument (e.g., QIAcuity plates). Integrated design simplifies loading and reduces cross-contamination risk [3] [23]. |
| Droplet Generation Oil & Cartridges | Reagents and consumables for creating a stable water-in-oil emulsion. | Critical for ddPCR workflow; droplet stability is essential for accurate partition counting [13]. |
| Nucleic Acid Extraction Kits | For purifying high-quality DNA or RNA from complex biological samples (e.g., plasma, swabs). | Automated platforms (e.g., KingFisher Flex, STARlet) are often used to ensure consistency and yield, which is critical for low-abundance targets [3] [13]. |
| qPCR-grade Water | A nuclease-free solvent for preparing reaction master mixes. | Prevents degradation of nucleic acids and reagents, ensuring reaction integrity. |
This guide provides an objective comparison of two principal readout methodologies in digital PCR (dPCR): in-line detection (exemplified by droplet digital PCR, or ddPCR) and planar imaging (exemplified by nanoplate-based dPCR). The focus is on their performance in generating precise and accurate data for research and drug development. While both methods provide absolute quantification of nucleic acids, their underlying technologies lead to distinct trade-offs in workflow, precision, and operational efficiency. Direct comparative studies reveal that both platforms are highly capable, but the choice between them often hinges on the specific requirements of the application, such as the need for maximum partitioning, workflow simplicity, or resistance to inhibitors.
The fundamental difference between in-line detection and planar imaging lies in how the PCR reaction is partitioned and analyzed.
- In-Line Detection (Droplet Digital PCR - ddPCR): This method uses a droplet generator to partition the PCR reaction mix into tens of thousands of nanoliter-sized, water-in-oil droplets. These droplets act as individual microreactors. After end-point PCR amplification in a thermal cycler, the droplets are streamed one-by-one past a optical detector in a flow cytometer-like setup—an "in-line" process—to determine the fluorescence of each droplet [6] [25]. The QX200 system from Bio-Rad is a classic example of this technology.
- Planar Imaging (Nanoplate Digital PCR - ndPCR): This method utilizes a microfluidic chip or nanoplate containing a fixed array of nanoliter-sized wells. The PCR mix is partitioned into these wells by a single loading step. The entire plate is then sealed, and PCR amplification occurs within the integrated instrument. Following amplification, the instrument captures a fluorescence image of the entire plate—a "planar imaging" process—and software analyzes the positive and negative wells [6] [8]. The QIAcuity system from QIAGEN is a leading platform using this approach.
The following workflow diagrams illustrate the distinct steps involved in each methodology.
Workflow: In-Line Detection (ddPCR)
Workflow: Planar Imaging (ndPCR)
Performance Comparison: Precision, Sensitivity, and Accuracy
Direct, side-by-side comparisons of these two methodologies are essential for informed decision-making. Recent studies have evaluated platforms like the QX200 ddPCR (in-line detection) and the QIAcuity ndPCR (planar imaging) across key performance metrics.
Table 1: Comparative Platform Performance from Recent Studies
Data synthesized from direct comparison studies [5] [8].
| Performance Metric | In-Line Detection (ddPCR) | Planar Imaging (ndPCR) | Context & Notes |
|---|---|---|---|
| Limit of Detection (LOD) | ~0.17 copies/µL input [8] | ~0.39 copies/µL input [8] | Measured using synthetic oligonucleotides. |
| Limit of Quantification (LOQ) | ~4.26 copies/µL input [8] | ~1.35 copies/µL input [8] | Determined by 3rd degree polynomial model fit. |
| Precision (Coefficient of Variation) | 6% to 13% [8] | 7% to 11% [8] | Across dilution series of synthetic DNA; both show high precision. |
| Accuracy vs. PFGE Gold Standard | 95% concordance [26] | Information not available in search results | PFGE (Pulsed-Field Gel Electrophoresis) for copy number variation. |
| Correlation between Platforms | R = 0.954 [5] | R = 0.954 [5] | Comparison of CDH13 gene methylation levels in breast cancer samples. |
Table 2: Technical and Operational Characteristics
Data compiled from technology reviews and manufacturer information [6] [8].
| Characteristic | In-Line Detection (ddPCR) | Planar Imaging (ndPCR) |
|---|---|---|
| Partitioning Method | Droplet generator [6] | Nanoplate [6] |
| Typical Number of Partitions | 20,000 (QX200) [6] | 8,500 - 26,000 (QIAcuity) [6] |
| Partition Volume | Picoliter (pL) scale [6] | Nanoliter (nL) scale [6] |
| Workflow | Discontinuous; multiple instruments (generator, cycler, reader) [6] | Integrated; single instrument (partitioning, cycling, imaging) [6] |
| Throughput (samples/run) | 96 (single 96-well plate) [6] | 24 to 96 (depending on plate format) [6] |
| Multiplexing Capacity | Up to 4 colors [6] | Up to 5 colors (plex) [6] |
| Susceptibility to "Rain" | Higher, due to droplet variability [6] | Lower [6] |
Experimental Protocols for Key Comparisons
To ensure reproducibility and provide context for the data in the tables above, here are summaries of the experimental methodologies used in the cited comparative studies.
- Objective: To compare the precision, sensitivity, and agreement of the QX200 ddPCR (in-line) and QIAcuity ndPCR (planar) platforms.
- Sample Material: DNA extracted from varying cell numbers of the ciliate Paramecium tetraurelia and synthetic oligonucleotides.
- Assay Details: Target gene copy number was quantified. The impact of two restriction enzymes (EcoRI and HaeIII) was tested.
- dPCR Workflow:
- ddPCR (QX200): Reactions were prepared in 20 µL volumes, droplets were generated, and PCR was performed in a C1000 thermal cycler. Droplets were read using a QX200 droplet reader.
- ndPCR (QIAcuity): Reactions were prepared in a 12 µL volume and pipetted into a 24-well nanoplate. The QIAcuity One instrument performed partitioning, thermocycling, and imaging automatically.
- Data Analysis: Limits of detection (LOD) and quantification (LOQ) were calculated. Precision was reported as the Coefficient of Variation (CV), and accuracy was assessed by comparing expected versus measured gene copies.
- Objective: To evaluate the efficacy of the QIAcuity (planar) and QX200 (in-line) dPCR platforms for sensitive detection of DNA methylation.
- Sample Material: 141 formalin-fixed, paraffin-embedded (FFPE) breast cancer tissue samples.
- Assay Details: Methylation-specific assay for the CDH13 gene promoter region using FAM-labeled probe for methylated and HEX-labeled probe for unmethylated DNA.
- dPCR Workflow:
- ndPCR (QIAcuity): 12 µL reactions were loaded into a nanoplate (8,500 partitions/well). Cycling and imaging were performed on the integrated QIAcuity One instrument.
- ddPCR (QX200): 20 µL reactions were loaded into a DG8 cartridge. Droplets were generated, transferred to a 96-well plate for PCR in a thermal cycler, and then read on a QX200 droplet reader.
- Data Analysis: Methylation level was calculated as the ratio of FAM-positive partitions to all positive partitions (FAM+HEX). Specificity and sensitivity were determined, and a correlation analysis between the two platforms was performed.
The Scientist's Toolkit: Essential Research Reagents
Successful execution of dPCR experiments, regardless of platform, relies on a set of core reagents. The following table details these essential components.
Table 3: Key Research Reagent Solutions for dPCR
Information derived from methods sections of comparative studies [5] [8] [27].
| Reagent / Material | Function | Example Products / Components |
|---|---|---|
| Digital PCR Master Mix | Provides optimized buffer, dNTPs, and polymerase for efficient amplification in partitions. | QIAcuity Probe PCR Master Mix [5], Supermix for Probes (No dUTP) for QX200 [5] [27] |
| Primers & Probes | Sequence-specific oligonucleotides for target amplification and detection. Hydrolysis (TaqMan) probes are common. | FAM- and HEX-labeled probes for target and reference/control [5] [8] |
| Restriction Enzymes | Used to digest high-molecular-weight DNA, improving access to the target sequence and accuracy of copy number estimation. | EcoRI, HaeIII [8] |
| DNA Modification Kits | For specific applications like bisulfite conversion, which distinguishes methylated from unmethylated cytosine in DNA. | EpiTect Bisulfite Kit [5] |
| Nucleic Acid Isolation Kits | For high-quality DNA extraction from various biological sources, including challenging samples like FFPE tissue. | DNeasy Blood and Tissue Kit [5] |
| Inhibitor Removal Kits | Critical for analyzing complex samples (e.g., wastewater, FFPE) by removing contaminants that inhibit PCR. | OneStep PCR Inhibitor Removal Kit [27] |
Both in-line detection (ddPCR) and planar imaging (ndPCR) are powerful tools for the absolute quantification of nucleic acids, offering superior precision and robustness compared to qPCR in many applications [26] [25]. The choice between them is not a matter of one being universally superior, but rather which is better suited to a laboratory's specific needs.
- In-line detection (ddPCR) traditionally generates a higher number of partitions, which can theoretically enhance precision and dynamic range. It is a well-established, highly sensitive method.
- Planar imaging (ndPCR) offers a significant advantage in workflow, integrating partitioning, amplification, and reading into a single instrument, which reduces hands-on time, minimizes risk of contamination, and simplifies operation [6]. Its insensitivity to "rain" can also facilitate data analysis [6].
For research and drug development environments where throughput, ease of use, and a streamlined workflow are paramount, planar imaging systems present a compelling option. For applications demanding the absolute maximum number of partitions, in-line detection remains a strong choice. Ultimately, the convergence of performance data between these platforms, as shown in direct comparisons, empowers scientists to select the tool that best aligns with their practical requirements for precision and efficiency.
dPCR in Action: Methodological Workflows and Sector-Specific Applications
Digital PCR (dPCR) represents a significant advancement in nucleic acid quantification by enabling absolute counting of target molecules without the need for standard curves. This technology operates by distributing a sample across thousands to millions of individual partitions, each serving as a miniature PCR reactor. After endpoint amplification, the proportion of positive partitions enables absolute quantification of the target sequence through Poisson statistics [6] [28]. While all dPCR platforms share this core principle, their implementation through partitioning strategies creates critical differences in workflow, precision, and practical application.
The two dominant partitioning strategies are droplet digital PCR (ddPCR), which uses water-in-oil emulsions to create partitions, and nanoplate-based dPCR, which employs microfluidic chips with fixed wells [6] [16]. This guide provides an objective, data-driven comparison of these technologies, focusing on their workflow implications for research and diagnostic applications.
Technology and Workflow Comparison
The fundamental difference between these platforms lies in their partitioning mechanisms and workflow integration, which directly impacts laboratory efficiency, reproducibility, and suitability for different environments.
Droplet Digital PCR (ddPCR) utilizes an immiscible fluid in oil to generate tens of thousands of submicroliter droplets that randomly encapsulate nucleic acids. A typical ddPCR workflow requires multiple instruments: a droplet generator for partition creation, a conventional thermocycler for endpoint PCR, and a flow cytometer-based droplet reader for fluorescence detection. This multi-step process involves significant manual intervention, including pipetting and transferring the emulsion between instruments [6] [16].
Nanoplate-based dPCR performs partitioning using a dedicated digital PCR plate containing 8,500 to 26,000 nanoscale wells. The key differentiator is full workflow integration: partitioning, thermocycling, and imaging all occur within a single instrument. The workflow resembles traditional qPCR—researchers pipette master mix and samples into the nanoplate, load it into the instrument, and retrieve results approximately two hours later with minimal hands-on time [6] [29] [30].
Table 1: Core Workflow Characteristics of ddPCR vs. Nanoplate dPCR
| Parameter | Droplet Digital PCR (ddPCR) | Nanoplate Digital PCR (ndPCR) |
|---|---|---|
| Partitioning Method | Water-oil emulsion droplets [6] [16] | Fixed micro-wells in a nanoplate [6] [16] |
| Workflow Integration | Multiple instruments (generator, thermocycler, reader) [6] | Single integrated instrument [6] [30] |
| Typical Time to Result | 6-8 hours [16] | Approximately 2 hours [29] [30] |
| Hands-on Time | High (multiple transfer steps) [6] | Low ("sample-in, results-out") [16] [30] |
| Primary Workflow Risk | Cross-contamination from transfers; droplet shearing/coalescence [6] | Minimal risk due to closed system [6] |
| Ideal Setting | Research and development labs [16] | Quality control (QC) and regulated environments [16] |
Workflow Visualization
The following diagrams illustrate the core procedural differences between the two technologies, highlighting the significant divergence in steps and component integration.
Diagram 1: Procedural workflows for ddPCR and ndPCR. The ddPCR process involves multiple instruments and transfer steps (red-themed), while the ndPCR workflow is consolidated within a single instrument (green-themed).
Diagram 2: System architecture comparison. ddPCR relies on a multi-instrument setup requiring manual sample transfer, whereas the ndPCR system integrates all modules into a single, automated unit.
Performance and Precision: Experimental Data
Independent studies have systematically compared the performance of droplet-based and nanoplate-based systems, providing empirical data on their precision, sensitivity, and reproducibility.
Comparative Platform Study on Gene Copy Number Quantification
A 2025 study directly compared the QX200 ddPCR system (Bio-Rad) and the QIAcuity One ndPCR system (QIAGEN) using synthetic oligonucleotides and DNA from the ciliate Paramecium tetraurelia to evaluate limits of detection, quantification, and precision [28].
- Methodology: Researchers performed absolute quantification of target genes using both platforms across a series of dilution levels. The study tested the impact of two restriction enzymes (EcoRI and HaeIII) on the precision of gene copy number measurements, especially for targets with potential tandem repeats. Data were analyzed according to the dMIQE guidelines to ensure robustness [28].
- Key Findings:
- Limit of Detection (LOD): The LOD for ndPCR was approximately 0.39 copies/µL input, while ddPCR showed a slightly lower LOD of 0.17 copies/µL input [28].
- Limit of Quantification (LOQ): The LOQ for ndPCR was determined to be 1.35 copies/µL input, which was lower than the ddPCR LOQ of 4.26 copies/µL input [28].
- Precision: Both platforms demonstrated high precision (Coefficient of Variation, CV, between 6-13%) across most concentrations above the LOQ. The choice of restriction enzyme significantly impacted ddPCR precision (CVs from 2.5% to 62.1% with EcoRI), whereas ndPCR showed consistently low CVs (0.6% - 6.5%) regardless of the enzyme used, indicating greater robustness [28].
Table 2: Performance Metrics from Comparative Study [28]
| Metric | QIAcuity ndPCR | QX200 ddPCR |
|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL | 0.17 copies/µL |
| Limit of Quantification (LOQ) | 1.35 copies/µL | 4.26 copies/µL |
| Precision (CV) - High Concentration | ~7-8% | ~6-7% |
| Precision (CV) - Low Concentration | ~11% | ~13% |
| Impact of Restriction Enzyme | Low (minimal CV change) | High (CV up to 62% with EcoRI) |
| Accuracy (vs. Expected Copies) | Consistently slightly lower | Consistently slightly lower, better agreement at mid-range |
GMO Quantification Validation Study
A 2025 in-house validation study for quantifying genetically modified soybeans further confirmed the equivalence of both platforms for precise quantitative analysis. The study validated duplex methods for the MON-04032-6 and MON89788 events with the lectin reference gene [20].
- Methodology: The validation followed JRC Guidance documents and involved testing specificity, dynamic range, linearity, limit of quantification, and accuracy. Identical primer-probe sets were used on both the QIAcuity and QX200 platforms to enable a direct comparison [20].
- Key Findings: The performance parameters for both duplex dPCR methods met all acceptance criteria on both platforms. The methods were equivalent in performance to the established singleplex qPCR method and deemed suitable for a full collaborative trial. This demonstrates that both ddPCR and ndPCR are fit-for-purpose for applications requiring high quantitative accuracy, such as regulatory compliance testing [20].
The Scientist's Toolkit: Essential Reagents and Materials
The following table details key reagents and consumables required for digital PCR experiments, drawing from the methodologies described in the cited studies.
Table 3: Essential Research Reagent Solutions for Digital PCR
| Item | Function | Application Notes |
|---|---|---|
| dPCR Master Mix | Provides enzymes, dNTPs, and buffer for amplification. | Critical performance factor. Choice of probe-based (e.g., for multiplexing) or EvaGreen dye-based mixes depends on application [28] [31]. |
| Primers & Probes | Target-specific oligonucleotides for amplification and detection. | Validated assays are essential for accuracy. ndPCR offers higher multiplexing capabilities (up to 12-plex) [6] [30]. |
| Restriction Enzymes | Digest DNA to reduce fragment size and improve access to targets. | Enhances precision, especially for complex or tandemly repeated genes. Enzyme choice (e.g., HaeIII vs. EcoRI) can significantly impact ddPCR results [28]. |
| Nanoplates | Microfluidic plates containing fixed partitions. | ndPCR consumable. Available in different well and partition number formats (e.g., 24- or 96-well, 26k partitions) [30]. |
| Droplet Generation Cartridges/Oil | Consumables for creating water-in-oil emulsion droplets. | ddPCR-specific consumables. Required for the initial partitioning step on systems like the QX200 [6]. |
| DNA Standards & Controls | Certified reference materials for assay validation. | Crucial for validating accuracy and trueness, especially in regulated applications like GMO testing [20]. |
Application Context: Choosing the Right Tool
The choice between ddPCR and ndPCR is often application- and context-dependent.
- ddPCR has a proven track record in research and is well-suited for assay development and environments where maximum partition numbers are desired. However, its multi-step workflow presents challenges in GMP and quality control settings where cross-contamination risks and operator error must be minimized [6] [16].
- ndPCR, with its integrated design and streamlined workflow, offers a compelling solution for routine testing, clinical diagnostics, and regulated environments. Its rapid time-to-result (under 2 hours) and minimal hands-on time enhance throughput and reduce operational costs in quality control labs [16] [30]. Studies have successfully applied ndPCR to diverse fields, including pathogen detection, gene expression analysis in cell and gene therapies, and sensitive copy number variation analysis [29] [30] [32].
Both droplet-based and nanoplate-based dPCR platforms provide excellent precision and accuracy for absolute nucleic acid quantification, outperforming qPCR in many applications [26] [32]. The core differentiator lies in their workflow and operational robustness.
The multi-step ddPCR workflow, while powerful, introduces complexity and variability through droplet handling and transfers. In contrast, integrated nanoplate systems offer a consolidated, "sample-to-result" workflow that minimizes manual intervention, reduces contamination risk, and provides faster results. For researchers prioritizing maximum workflow efficiency, reproducibility, and suitability for regulated clinical or quality control environments, nanoplate-based dPCR presents a significant operational advancement. For discovery research requiring the highest possible partition count, droplet-based methods remain a valuable tool. The decision ultimately hinges on the specific requirements for precision, throughput, and operational simplicity within the intended application.
Digital PCR (dPCR) has emerged as a powerful third-generation PCR technology for the absolute quantification of nucleic acids, revolutionizing the field of oncology by enabling non-invasive liquid biopsy analysis and the detection of ultra-rare mutations. [2] By partitioning a PCR reaction into thousands of individual reactions, dPCR allows for the detection and precise quantification of circulating tumor DNA (ctDNA) present in patient blood samples at extremely low frequencies, often below 0.1% variant allele frequency. [33] This sensitivity is crucial for monitoring treatment response, detecting minimal residual disease (MRD), and identifying emerging resistance mutations during targeted therapy. [34] Two primary dPCR platforms have dominated the research landscape: droplet digital PCR (ddPCR) systems, which encapsulate reactions in water-in-oil emulsions, and nanoplate-based dPCR systems, which utilize microchambers embedded in solid chips. [28] [2] This guide provides an objective comparison of these competing technologies, focusing on their performance characteristics, experimental parameters, and applicability to oncological research and clinical development.
Technology Comparison: Nanoplate vs. Droplet Digital PCR
Core Principles and Technical Specifications
Droplet Digital PCR (ddPCR) partitions PCR reactions into thousands of nanoliter-sized water-in-oil droplets using microfluidic chips. [2] The QX200 system from Bio-Rad is a prominent example, generating up to 20,000 droplets per sample. After end-point PCR amplification, droplets flow single-file through a detector that reads fluorescence, classifying each as positive or negative. [28] The absolute concentration of the target molecule is then calculated using Poisson statistics.
Nanoplate Digital PCR (ndPCR), exemplified by the QIAcuity system from QIAGEN, performs partitioning through microfluidic circuits that distribute the reaction mix into nanoscale chambers etched into a plastic chip. [28] [2] These systems typically utilize integrated thermocyclers and perform fluorescence detection via imaging of the entire plate at once, eliminating the need for droplet flow control.
Table 1: Technical Comparison of Representative dPCR Platforms
| Feature | Nanoplate dPCR (QIAcuity One) | Droplet dPCR (QX200) |
|---|---|---|
| Partitioning Mechanism | Solid-state microchambers [28] | Water-in-oil droplets [2] |
| Typical Partition Count | Not explicitly stated (fewer but larger volume) | Up to 20,000 droplets [2] |
| Reaction Volume | 40 µL [28] | 20 µL [28] |
| Detection Method | Planar fluorescence imaging [28] | In-line droplet fluorescence detection [2] |
| Multiplexing Capacity | 5-color detection [34] | 2-color standard (upgradable) [34] |
| Sample Throughput | Higher (integrated, automated plating) [34] | Lower (manual droplet generation) [35] |
| Key Advantage | Ease of use, automation, reduced risk of contamination [2] | High partition count, established validation data [2] |
Performance Metrics in Liquid Biopsy Applications
Direct comparative studies reveal nuanced performance differences between the two platforms, particularly in clinical oncology settings. A 2023 study published in Clinica Chimica Acta directly compared a droplet-based system (Bio-Rad's ddPCR) and a solid-phase system (QIAGEN's QIAcuity) for analyzing liquid biopsy samples from lung and colorectal cancer patients. [4]
Table 2: Clinical Performance Comparison for Liquid Biopsy Analysis
| Metric | Nanoplate dPCR (QIAcuity) | Droplet dPCR (QX200) |
|---|---|---|
| EGFR Mutation Detection Rate | 100% (vs. tissue) [4] | 58.8% (vs. tissue) [4] |
| RAS Mutation Detection Rate | 86.4% (vs. tissue) [4] | 72.7% (vs. tissue) [4] |
| Overall Agreement (κ statistic) | Moderate agreement between platforms (κ = 0.34-0.54) [4] | Moderate agreement between platforms (κ = 0.34-0.54) [4] |
| Reported Sensitivity | Can detect mutations at 0.1% VAF in a single well [33] | Can detect mutations at 0.1% VAF [34] |
| Precision with ciliate DNA (CV with HaeIII enzyme) | < 5% CV [28] | < 5% CV [28] |
The study concluded that while there was moderate agreement between the platforms, the solid dPCR (QIAcuity) demonstrated a higher sensitivity in detecting mutated cases from plasma cfDNA compared to tissue results. [4] This suggests that platform choice can significantly impact clinical detection rates.
Experimental Protocols and Workflows
Standardized Workflow for Liquid Biopsy Analysis
The general workflow for detecting oncogenic mutations from liquid biopsies is similar across platforms but involves specific reagents and steps tailored to each system.
Detailed Methodologies from Cited Studies
1. Protocol for ctDNA Detection in Rectal Cancer (ddPCR) [14]:
- Sample Collection: Pre-therapy plasma and matched rectal tumor samples were collected from 41 patients in a development group.
- Mutation Identification: Tumor DNA was first sequenced via Next-Generation Sequencing (NGS) to identify patient-specific mutations.
- Assay Design: Custom ddPCR assays were designed against the identified mutations.
- ddPCR Setup: The reaction mixture was prepared using a ddPCR supermix, primers/probes, and plasma-derived cfDNA. The mixture was loaded into a droplet generator cartridge alongside droplet generation oil.
- Partitioning & Amplification: The QX200 droplet generator created ~20,000 droplets per sample, which were then transferred to a 96-well plate and sealed. PCR amplification was performed in a thermal cycler.
- Data Analysis: The QX200 droplet reader counted positive and negative droplets, and the mutant allele frequency was calculated using Poisson statistics.
2. Protocol for EGFR/KRAS Mutation Detection (ddPCR vs. ndPCR) [4]:
- Sample Preparation: Plasma samples from NSCLC and CRC patients were collected. cfDNA was extracted using commercial kits.
- Reaction Setup for QIAcuity (ndPCR): The reaction mix contained QIAcuity MasterMix, the respective mutation assay (e.g., dPCR LNA Mutation Assay), and template cfDNA. The mix was loaded into a QIAcuity Nanoplate, which was then sealed and placed in the instrument.
- Reaction Setup for QX200 (ddPCR): A similar reaction mix was prepared using ddPCR Supermix and loaded for droplet generation.
- Integrated vs. Separate Steps: The QIAcuity instrument performed partitioning, thermal cycling, and imaging automatically within the same instrument. For the QX200, droplet generation, PCR amplification, and droplet reading were separate, manual steps.
- Analysis: Both platforms provided absolute quantification of mutant and wild-type alleles. The QIAcuity software suite directly calculated the fractional abundance of the mutant target.
3. Key Consideration: Impact of Restriction Enzymes [28]: A methodological study comparing platforms highlighted that the choice of restriction enzyme can significantly impact precision, especially for ddPCR. Using HaeIII instead of EcoRI increased precision for the QX200 system, reducing the Coefficient of Variation (CV) to below 5% for both platforms when analyzing DNA from Paramecium tetraurelia. This step is often incorporated to digest longer DNA fragments and improve access to the target sequence.
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of dPCR in oncology research requires specific, high-quality reagents and materials. The following table details key components for a typical workflow.
Table 3: Essential Research Reagent Solutions for dPCR in Oncology
| Item | Function/Description | Example Application |
|---|---|---|
| dPCR Master Mix | A ready-to-use mix containing DNA polymerase, dNTPs, buffers, and stabilizers optimized for specific platforms. | QIAcuity MasterMix for nanoplate systems; ddPCR Supermix for droplet systems. [33] [4] |
| Mutation-Specific Assays | Primer and probe sets designed to selectively amplify and detect a specific mutant allele. Often use chemically modified nucleotides like LNA to enhance specificity. | dPCR LNA Mutation Assays for detecting oncogenic mutations in EGFR or KRAS at frequencies as low as 0.1%. [33] |
| Nanoplates or Cartridges | Disposable plastic chips containing microfluidic circuits and pre-defined chambers for reaction partitioning. | QIAcuity Nanoplate for integrated partitioning and amplification. [28] |
| Droplet Generation Oil & Cartridges | Specialized oil and surfactant stabilizes water-in-oil emulsions during droplet formation. | DG Cartridges and Droplet Generation Oil for the QX200 system. [14] |
| cfDNA Extraction Kits | Kits optimized for isolating short-fragment, low-concentration cell-free DNA from plasma samples. | QIAamp Viral RNA Mini Kit or INSTANT virus RNA/DNA kit, adapted for DNA extraction. [35] |
| Negative & Positive Controls | Certified DNA samples with known mutation status (positive) and nuclease-free water or wild-type DNA (negative) for assay validation. | Synthetic oligonucleotides with known sequences; DNA from characterized cell lines. [28] |
Both nanoplate and droplet digital PCR platforms offer exceptional sensitivity and precision for liquid biopsy applications in oncology, capable of detecting rare mutations down to 0.1% variant allele frequency. [33] The choice between them involves a trade-off between workflow efficiency and analytical depth. Nanoplate systems (e.g., QIAcuity) provide a more automated, integrated, and user-friendly workflow with higher multiplexing capacity, showing superior clinical detection rates in some direct comparisons. [4] [34] Droplet systems (e.g., QX200) offer a high number of partitions and a long history of validation data, making them a robust and established choice. [2]
The ongoing integration of artificial intelligence for automated droplet classification and data analysis is set to further enhance the throughput and accuracy of both platforms. [34] As the field moves toward standardized, reimbursable clinical diagnostics, understanding these nuanced performance and workflow differences is crucial for researchers and drug development professionals to select the optimal tool for their specific oncological application.
The accurate quantification of pathogenic microorganisms and the monitoring of viral loads are cornerstones of modern infectious disease management, influencing diagnostics, treatment efficacy assessment, and public health surveillance [36] [3]. While real-time quantitative PCR (qPCR) has long been the standard, digital PCR (dPCR) technologies have emerged as powerful tools offering absolute quantification without the need for standard curves [37] [38]. Among dPCR platforms, a key distinction exists between droplet digital PCR (ddPCR), which partitions samples into water-in-oil emulsion droplets, and nanoplate digital PCR (ndPCR), which distributes samples into fixed nanoscale wells on a microfluidic chip [16] [20]. This guide objectively compares the performance of these two platforms within the context of infectious disease research and diagnostics, framing the analysis within the broader thesis that precision and accuracy are paramount for reliable pathogen quantification. The comparison is grounded in recently published experimental data and validation studies.
The fundamental principle of digital PCR involves partitioning a PCR reaction into thousands of individual reactions, allowing for the absolute quantification of nucleic acid targets via Poisson statistics [8] [39]. Despite this shared principle, the partitioning mechanism constitutes the primary difference between ddPCR and ndPCR, leading to distinct practical workflows.
Droplet Digital PCR (ddPCR) employs a water-oil emulsion to generate typically 20,000 nanolitre-sized droplets, which act as independent reaction chambers [16]. This process involves several instruments: a droplet generator, a thermocycler for end-point PCR, and a droplet reader for fluorescence detection [20]. The multi-step, multi-instrument workflow is often cited as more time-consuming and hands-on.
In contrast, Nanoplate Digital PCR (ndPCR), as exemplified by systems like the QIAcuity, uses integrated microfluidic chips (nanoplates) containing a fixed array of nanoscale wells [8] [20]. This platform integrates partitioning, thermocycling, and imaging into a single, automated instrument, streamlining the process into a "sample-in, results-out" workflow that reduces hands-on time and potential for human error [16] [3].
The following diagram illustrates the core workflows for both technologies, highlighting the key procedural differences.
Performance Comparison: Quantitative Data
Direct comparative studies provide the most robust basis for evaluating the two technologies. The following table summarizes key performance metrics from recent research, including studies on pathogen detection and general platform comparisons.
Table 1: Comparative Performance Metrics of ddPCR and ndPCR
| Performance Parameter | Droplet Digital PCR (ddPCR) | Nanoplate Digital PCR (ndPCR) | Context and Observations |
|---|---|---|---|
| Limit of Detection (LOD) | 0.17 copies/µL input [8]1.99 copies/µL (SARS-CoV-2 N1) [36] | 0.39 copies/µL input [8]1.83 copies/µL (Canine Coronavirus) [38] | Both platforms offer high sensitivity, suitable for low-level viremia or early infection. Specific LOD is target-dependent [8] [38]. |
| Limit of Quantification (LOQ) | 4.26 copies/µL input [8]85.2 copies/reaction [8] | 1.35 copies/µL input [8]54 copies/reaction [8] | ndPCR demonstrated a lower LOQ in a direct platform comparison [8]. |
| Precision (as % Coefficient of Variation) | 6% - 13% (with synthetic oligos) [8]CV < 5% (with optimal restriction enzyme) [8] | 7% - 11% (with synthetic oligos) [8]Generally high and consistent precision [8] | Precision can be affected by sample type and preparation. Enzyme choice (e.g., HaeIII) can significantly improve ddPCR precision [8]. |
| Linear Range | R²: 0.999 (SARS-CoV-2 N1) [36]R²: 0.99 (synthetic oligos) [8] | R²: 0.98 (synthetic oligos) [8] | Both platforms exhibit excellent linearity across a wide dynamic range, enabling reliable quantification from low to high target concentrations [8] [36]. |
| Accuracy | Measured copies consistently lower than expected, but high agreement (R²adj=0.99) [8] | Measured copies consistently lower than expected, with high agreement (R²adj=0.98) [8] | Both platforms showed a slight underestimation bias in a controlled study, but maintained high correlation with expected values [8]. |
| Sensitivity vs. qPCR | 100 CFU/mL for L. casei (vs. lower for qPCR) [37] | 100-fold greater than RT-qPCR for Canine Coronavirus [38] | dPCR platforms generally outperform qPCR in sensitivity, particularly for complex samples or low viral loads [37] [3] [38]. |
Experimental Protocols for Infectious Disease Applications
The methodologies from cited studies provide validated protocols for implementing these technologies in pathogen detection.
Protocol: SARS-CoV-2 Detection and Quantification using ddPCR
This protocol is adapted from a study evaluating the clinical performance of ddPCR for SARS-CoV-2 detection [36].
- RNA Extraction: Extract viral RNA from nasopharyngeal swabs using a validated automated extraction system (e.g., KingFisher Flex, MagMAX Viral/Pathogen kit).
- Reverse Transcription and ddPCR Setup:
- Use a one-step or two-step RT-ddPCR approach with a commercial SARS-CoV-2 ddPCR kit (e.g., Bio-Rad SARS-CoV-2 ddPCR kit).
- Prepare a reaction mixture containing ddPCR Supermix for Probes (No dUTP), reverse transcriptase, primers and probes for SARS-CoV-2 targets (e.g., N1, N2), and the extracted RNA template.
- Droplet Generation: Transfer the reaction mixture to a DG8 cartridge with droplet generation oil. Generate droplets using a QX200 Droplet Generator.
- PCR Amplification: Carefully transfer the emulsified droplets to a 96-well PCR plate, seal the plate, and perform endpoint PCR on a thermal cycler. A typical cycling condition is: 95°C for 10 min (reverse transcription/activation), 40 cycles of 94°C for 30 sec and 55-60°C for 60 sec, followed by 98°C for 10 min and a 4°C hold.
- Droplet Reading and Analysis: Place the plate in a QX200 Droplet Reader, which counts the positive (fluorescent) and negative (non-fluorescent) droplets for each sample. The absolute concentration (copies/µL) is calculated by the instrument software (QuantaSoft) using Poisson statistics.
Protocol: Canine Respiratory Coronavirus Detection using ndPCR
This protocol is based on the development and validation of a nanoplate-based RT-dPCR assay [38].
- Primer/Probe Design: Design primers and a hydrolysis probe (e.g., 5'-end labeled with 6-FAM) targeting a conserved region of the pathogen's genome (e.g., the spike gene for CRCoV). Validate specificity in silico.
- RNA Extraction and Purification: Extract RNA from clinical swabs (nasal, oropharyngeal, rectal) using a viral RNA kit (e.g., QIAamp Viral RNA Mini kit) on an automated system (e.g., QIAcube Connect).
- ndPCR Reaction Setup:
- Use a one-step RT-dPCR reaction mix (e.g., QIAcuity OneStep Advanced Probe RT-PCR Kit).
- Prepare a master mix containing the reaction buffer, enzymes, optimized concentrations of primers and probe, and the RNA template.
- Loading and Integrated Run: Pipette the reaction mix into the wells of a QIAcuity nanoplate (e.g., 26k partition size). Seal the nanoplate and load it into the QIAcuity One instrument.
- Integrated Analysis: The instrument automatically performs partitioning into nanowells, thermocycling, and imaging. The QIAcuity Suite Software analyzes the fluorescence data in each nanowell, applies a threshold to distinguish positive from negative partitions, and provides an absolute copy number concentration (copies/µL) based on Poisson distribution.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of dPCR assays requires a suite of reliable reagents and materials. The following table details key solutions used in the featured studies.
Table 2: Essential Reagents and Materials for dPCR Assays in Pathogen Detection
| Item | Function | Examples from Literature |
|---|---|---|
| Digital PCR Supermix | Provides optimized buffer, dNTPs, and polymerase for robust amplification in partitioned reactions. | ddPCR Supermix for Probes (Bio-Rad) [39], QIAcuity Multiplex PCR Kit (QIAGEN) [20]. |
| Primers & Hydrolysis Probes | Ensure specific amplification and detection of the target pathogen gene. Must be highly specific and efficient. | Primers/Probes for SARS-CoV-2 N1/N2 [36], CRCoV spike gene [38], Salmonella ttr, invA genes [39]. |
| Nucleic Acid Extraction Kit | Isolate high-quality, inhibitor-free DNA or RNA from complex clinical samples (swabs, sputum, etc.). | MagMax Viral/Pathogen Kit [3], QIAamp Viral RNA Mini Kit [38], RSC PureFood GMO Kit [20]. |
| Restriction Enzymes | Can be used to digest genomic DNA and improve accessibility of tandemly repeated or complex targets, enhancing precision [8]. | HaeIII, EcoRI [8]. |
| Reference Materials | Certified standards used for assay validation, determining limits of detection, and ensuring accuracy. | AccuPlex SARS-CoV-2 Reference Material [36], ERM Certified Reference Materials [20]. |
| Consumables | Platform-specific partitions for reaction setup. | DG8 Cartridges & Droplet Generation Oil (Bio-Rad ddPCR) [39], QIAcuity Nanoplates (QIAGEN ndPCR) [20] [38]. |
Discussion and Pathogen-Specific Applications
The choice between ddPCR and ndPCR often depends on the specific pathogen and research question. The following diagram and discussion highlight key application areas and technology selection factors.
- Respiratory Virus Detection: During the 2023-2024 "tripledemic," dPCR demonstrated superior accuracy for quantifying influenza A, influenza B, RSV, and SARS-CoV-2 compared to RT-qPCR, especially at medium and high viral loads [3]. This precise quantification is crucial for understanding infection dynamics and co-infections.
- Coronavirus Applications: A nanoplate RT-dPCR assay for Canine Respiratory Coronavirus (CRCoV) showed 100-fold greater sensitivity than RT-qPCR, proving particularly effective for detecting low viral loads in rectal swabs where RT-qPCR underperformed [38].
- Bacterial Pathogen Quantification: ddPCR has been successfully validated for absolute quantification of Salmonella spp. with high specificity, precision (5-10% RSD), and a low LOD (0.5 copies/µL), making it suitable for food safety testing and public health surveillance [39].
- Complex Samples and Inhibitor Tolerance: The partitioning process in both ddPCR and ndPCR dilutes inhibitors present in complex sample matrices (e.g., sputum, food samples), making these techniques more robust than qPCR [37] [3] [39]. This is a significant advantage for direct analysis of clinical and environmental samples.
Both droplet digital PCR and nanoplate digital PCR offer significant advantages over traditional qPCR for pathogen quantification and viral load monitoring, primarily through absolute quantification, high sensitivity, and robustness to inhibitors. The choice between the two depends on specific application needs.
- Nanoplate dPCR (ndPCR) presents a compelling case for laboratories prioritizing a streamlined, automated workflow with high throughput and consistently high precision, making it highly suitable for clinical quality control environments and standardized testing [16] [20] [3]. Recent studies also indicate its potential for superior sensitivity in certain applications [38].
- Droplet dPCR (ddPCR) remains a well-established, highly validated technology with an extensive body of literature supporting its use. It is an excellent choice for research settings requiring proven high-plexing capabilities or for applications where established protocols are paramount [36] [16] [39].
The broader thesis on precision and accuracy affirms that both platforms are highly capable. The decision should be guided by a fit-for-purpose evaluation, considering factors such as required throughput, workflow integration, the need for ultra-sensitive detection, and the specific pathogenic targets of interest.
Application in Food Authentication and GMO Quantification
In the fields of food authentication and Genetically Modified Organism (GMO) quantification, the demand for precise and reliable molecular detection methods has never been greater. Regulatory frameworks, such as the European Union's requirement to label food and feed containing more than 0.9% of approved GMOs per ingredient, create an imperative for analytical techniques capable of accurate quantification at low thresholds [20]. Similarly, the detection of species adulteration in meat products addresses critical concerns regarding food fraud, allergen management, and religious dietary compliance [19]. For over a decade, real-time quantitative PCR (qPCR) has served as the gold standard for nucleic acid-based detection in these areas. However, the emergence of digital PCR (dPCR) represents a significant technological shift, offering absolute quantification without the need for standard curves and demonstrating enhanced resilience to PCR inhibitors [20] [40].
Digital PCR technology has diversified into two primary partitioning methodologies: droplet digital PCR (ddPCR), which employs a water-oil emulsion to generate thousands of nanoliter-sized droplets, and nanoplate-based digital PCR (ndPCR), which distrib samples across a microfluidic chip containing fixed nanowells [2] [16]. This guide provides an objective, data-driven comparison of these platforms, focusing on their performance in food authentication and GMO quantification, framed within the broader thesis that precision and accuracy are paramount in analytical food science.
Technology Comparison: Partitioning Mechanisms and Workflows
The fundamental difference between ddPCR and ndPCR lies in their partitioning mechanisms and associated workflows, which directly influence their ease of use, reproducibility, and suitability for different laboratory environments.
Workflow and Practical Handling
Droplet Digital PCR (ddPCR) utilizes a multi-step workflow that involves preparing a reaction mixture, transferring it to a droplet generation cartridge, generating droplets in a water-oil emulsion, manually transferring the emulsified droplets to a PCR plate, performing thermocycling, and finally reading the droplets in a droplet reader [20] [16]. This process involves multiple instruments and manual transfer steps, which can increase hands-on time and the potential for cross-contamination or droplet shearing [19] [16].
In contrast, Nanoplate Digital PCR (ndPCR) streamlines the process into a "sample-in, results-out" workflow. The reaction mixture is loaded into a dedicated nanoplate, which is then sealed and placed into a fully integrated instrument. This instrument automatically performs partitioning, thermocycling, and imaging without the need for further manual intervention [20] [16]. This integrated system reduces hands-on time, minimizes the risk of contamination, and offers a workflow more familiar to users of traditional qPCR [19].
The diagram below illustrates the core operational difference between the two partitioning methods.
Performance Data in Food Authentication and GMO Analysis
Robust experimental data from peer-reviewed studies allows for a direct comparison of the technical performance of ddPCR and ndPCR in applied settings.
Performance in GMO Quantification
A 2025 study directly compared the Bio-Rad QX200 ddPCR and Qiagen QIAcuity ndPCR platforms for the quantification of two major GM soybean events (MON-04032-6 and MON89788) using identical primer-probe sets. The in-house validation assessed critical parameters including dynamic range, linearity, limit of quantification, and accuracy [20].
- Accuracy and Validation: The study concluded that both platforms demonstrated performance that agreed with acceptance criteria from JRC Guidance documents. The duplex dPCR methods were found to be equivalent in performance to singleplex qPCR methods and deemed suitable for collaborative trials towards full validation [20].
- Practical Application: Another study highlighted dPCR's particular utility for analyzing complex food matrices, where qPCR can struggle. It was shown that complex real-life samples which could not be quantified by qPCR were successfully quantified using ddPCR, underscoring its robustness [40].
Performance in Food Authentication
A 2025 study developed a novel duplex ndPCR assay for the simultaneous detection of pork and chicken in processed meat products, targeting the mitochondrial D-loop region and cytochrome b gene [19].
- Sensitivity and Limit of Detection (LOD): The ndPCR assay demonstrated a limit of detection of 0.1% (w/w) for both pork and chicken in beef. This was reported to be more sensitive than the 1% LOD achieved with real-time PCR in the same study, highlighting the superior sensitivity of the nanoplate dPCR method for food authentication [19].
- Applicability: The assay was successfully applied to 27 commercial meat products, with results consistent with label information, proving its practical utility for food fraud detection [19].
Further demonstrating the sensitivity of ndPCR, a 2024 study developed the first nanoplate dPCR method for tracing allergenic foods, specifically sesame. The method achieved sensitivities of 5 mg/kg and 0.1 mg/kg in dough and biscuit matrices, improving upon real-time PCR sensitivity by one order of magnitude. The performance was not affected by food processing or complex matrices [22].
Comparative Platform Precision
A 2025 study provided a direct, cross-platform performance comparison using synthetic oligonucleotides and DNA from the ciliate Paramecium tetraurelia [8].
- Precision and Robustness: The study found that both the QX200 ddPCR and QIAcuity ndPCR platforms showed high precision across most analyses. The coefficient of variation (CV) for both platforms indicated precise results, with CVs ranging between 6-13% for ddPCR and 7-11% for ndPCR for synthetic oligonucleotides. The study also highlighted that the choice of restriction enzyme could impact precision, particularly for the ddPCR system, whereas ndPCR showed less variability based on enzyme choice [8].
- Limits of Detection and Quantification: The study determined that the LOD for ndPCR was approximately 0.39 copies/µL input, while for ddPCR it was 0.17 copies/µL input. Conversely, the Limit of Quantification (LOQ) for ndPCR was 1.35 copies/µL input, which was lower than the 4.26 copies/µL input for ddPCR in this specific experimental setup [8].
The table below summarizes key performance metrics from the cited studies.
Table 1: Comparative Performance Metrics of ddPCR and ndPCR
| Application | Performance Metric | ddPCR (Bio-Rad QX200) | ndPCR (Qiagen QIAcuity) | Citation |
|---|---|---|---|---|
| GMO Quantification | Validation Outcome | Meets acceptance criteria | Meets acceptance criteria | [20] |
| Meat Authentication | Limit of Detection (LOD) | Not specified in context | 0.1% (w/w) for pork/chicken | [19] |
| Allergen Detection | Sensitivity in Food Matrix | Not specified in context | 0.1 - 5 mg/kg of sesame | [22] |
| Gene Copy Analysis | Limit of Detection (LOD) | ~0.17 copies/µL | ~0.39 copies/µL | [8] |
| Gene Copy Analysis | Limit of Quantification (LOQ) | ~4.26 copies/µL | ~1.35 copies/µL | [8] |
| Gene Copy Analysis | Precision (Coefficient of Variation) | 6% - 13% | 7% - 11% | [8] |
Detailed Experimental Protocols
To ensure reproducibility and provide a clear understanding of the underlying data, this section outlines the key experimental methodologies from the core comparative studies.
Protocol for Duplex GMO Quantification
This protocol is adapted from the 2025 comparison study for quantifying MON-04032-6 and MON89788 soybean events [20].
- DNA Extraction: Extract genomic DNA from certified reference materials (CRMs) using a validated kit, such as the RSC PureFood GMO kit with a Maxwell RSC Instrument, or a CTAB-based method as described in ISO 21571:2005.
- DNA Quality Assessment: Quantify the DNA concentration and assess purity. Perform an inhibition test by measuring the copy number of a reference gene (e.g., lectin for soybean) at three serial dilution levels in duplicate. The measured copies in diluted samples should not differ by more than 25% from the highest concentration.
- Sample Preparation: Prepare desired GM percentage levels (e.g., 10%, 2%, 0.5%) by gravimetrically mixing GM and non-GM materials, considering the absolute copy number of the reference gene.
- dPCR Reaction Setup:
- For QX200 ddPCR: Prepare a duplex reaction mixture containing primers and probes for the GM event and the reference gene, along with ddPCR Supermix for Probes, and DNA template. Generate droplets using a QX200 Droplet Generator.
- For QIAcuity ndPCR: Prepare a similar duplex reaction mixture using the QIAcuity Probe PCR Kit. Load the mixture into a QIAcuity Nanoplate 26k.
- Thermocycling and Analysis: Perform endpoint PCR amplification. Read the droplets on a QX200 Droplet Reader or image the nanoplate in the integrated QIAcuity instrument. Analyze data using manufacturer-specific software (QX Manager or QIAcuity Suite) to obtain absolute copy numbers per microliter.
Protocol for Meat Authentication via Duplex ndPCR
This protocol is based on the 2025 study for simultaneous detection of pork and chicken [19].
- Sample Collection and DNA Extraction: Collect meat samples from target and non-target species. Extract total DNA from raw and processed meat products using a commercial DNA extraction kit suitable for complex matrices.
- Primer/Probe Design: Design and validate primer-probe sets specific to the target species. The referenced study targeted the mitochondrial D-loop region (pork) and the cytochrome b gene (chicken).
- Nanoplate dPCR Setup:
- Prepare the duplex PCR reaction mixture containing the respective primers and probes for both targets, the QIAcuity Probe PCR Master Mix, and the extracted DNA template.
- Load the mixture into a QIAcuity nanoplate.
- Partitioning and Amplification: Place the sealed nanoplate into the QIAcuity instrument. The system automatically performs partitioning into ~26,000 wells, PCR amplification, and endpoint fluorescence imaging.
- Data Analysis: Use the QIAcuity Suite Software to analyze the fluorescence data. The software automatically calculates the absolute copy number of each target per microliter of reaction, allowing for the determination of the presence and proportion of each species.
Essential Research Reagent Solutions
The following table catalogues key reagents and materials required for implementing dPCR assays in food analysis, as derived from the experimental protocols in the search results.
Table 2: Key Research Reagents and Materials for dPCR-based Food Analysis
| Reagent / Material | Function | Example Products / Components |
|---|---|---|
| Certified Reference Materials (CRMs) | Provides calibrated standards for GMO quantification and method validation. | MON-04032-6 SOYA BEAN ERM-BF410 series; MON89788 from AOCS [20] |
| DNA Extraction Kits | Iserts high-quality, inhibitor-free genomic DNA from complex food matrices. | RSC PureFood GMO Kit; CTAB buffer method; MagMax Viral/Pathogen Kit [20] [3] |
| dPCR Master Mix | Provides optimized buffers, polymerase, and nucleotides for efficient amplification in partitions. | ddPCR Supermix for Probes (Bio-Rad); QIAcuity Probe PCR Kit (QIAGEN) [20] [31] |
| Primer/Probe Sets | Confers specificity for target DNA sequences (GM events, species-specific genes, allergens). | Event-specific assays for MON-04032-6/MON89788; mitochondrial D-loop/cyt b probes [20] [19] |
| Partitioning Consumables | Creates the nanoreactions for absolute quantification. | DG8 Cartridges & Droplet Generation Oil (ddPCR); QIAcuity Nanoplate 26k (ndPCR) [20] [19] |
The collective experimental data demonstrates that both droplet-based and nanoplate-based digital PCR platforms are highly capable technologies for sensitive and precise analysis in food authentication and GMO quantification. Both platforms outperform qPCR in aspects of sensitivity and robustness to inhibitors [20] [19] [40].
The choice between ddPCR and ndPCR often hinges on specific laboratory priorities. ddPCR platforms have a long track record and are well-established in research settings. ndPCR platforms, with their integrated, automated workflows, offer significant advantages in operational efficiency, reduced hands-on time, and a lower risk of contamination, making them particularly suited for quality control environments and high-throughput laboratories [16]. The performance data indicates that ndPCR consistently achieves detection limits that meet or exceed regulatory requirements which are technically challenging for qPCR, such as the 0.1% meat adulteration level and the 0.9% GMO labeling threshold [20] [19].
Ultimately, the decision is fit-for-purpose. For laboratories where throughput, ease of use, and integration into standardized workflows are paramount, ndPCR presents a compelling solution. For research applications requiring maximum flexibility or building upon an established ddPCR infrastructure, ddPCR remains a powerful and validated tool. Both technologies unequivocally advance the thesis that precision and accuracy in food analysis are best served by the absolute quantification capabilities of digital PCR.
Digital PCR (dPCR) has emerged as a pivotal technology for cell and gene therapy manufacturing, providing absolute quantification of nucleic acids without the need for standard curves. This capability is particularly valuable for critical quality control (QC) applications such as vector copy number (VCN) determination and release testing, where precision and accuracy are paramount [41]. The two principal dPCR methodologies—nanoplate-based digital PCR (ndPCR) and droplet digital PCR (ddPCR)—differ fundamentally in their approach to sample partitioning, which in turn influences their performance characteristics and suitability for regulated manufacturing environments [6] [16].
In ndPCR, reactions are partitioned into fixed nanowells on a microfluidic chip, with all processes integrated into a single instrument. In contrast, ddPCR relies on generating thousands of nanoliter-sized droplets through water-oil emulsion, often requiring multiple instruments for droplet generation, thermocycling, and reading [6] [42]. This comparison guide objectively evaluates the performance of these platforms within the context of cell and gene therapy, focusing on experimental data, precision, and workflow efficiency to inform platform selection for critical QC assays.
Technology Comparison: Performance and Operational Characteristics
Quantitative Performance Metrics
Direct comparative studies reveal nuanced performance differences between nanoplate and droplet dPCR systems. A 2025 study comparing the QIAcuity One (ndPCR) and QX200 (ddPCR) platforms using synthetic oligonucleotides and ciliate DNA demonstrated both platforms achieved high precision, though their limits of detection and quantification varied slightly [8].
Table 1: Performance Metrics from Comparative Studies
| Performance Parameter | Nanoplate dPCR (QIAcuity) | Droplet ddPCR (QX200) |
|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL input [8] | 0.17 copies/µL input [8] |
| Limit of Quantification (LOQ) | 1.35 copies/µL input [8] | 4.26 copies/µL input [8] |
| Accuracy (vs. expected copies) | R²adj = 0.98 [8] | R²adj = 0.99 [8] |
| Typical Partitions | 8,500 or 26,000 [6] | 20,000 [6] |
| Multiplexing Capability | Up to 5-plex [6] | Up to 4-plex (QX ONE) [6] |
The study found restriction enzyme selection significantly impacted precision, particularly for ddPCR. When using HaeIII instead of EcoRI, ddPCR precision improved markedly, with all coefficients of variation (CVs) falling below 5%. This effect was less pronounced for ndPCR, which maintained more consistent precision across enzyme choices [8]. Both platforms demonstrated linear quantification of gene copies across increasing cell numbers, confirming their reliability for absolute quantification applications in biological samples [8].
Operational and Workflow Characteristics
Operational considerations significantly impact platform selection for GMP environments where efficiency, reproducibility, and compliance are critical.
Table 2: Operational Comparison of dPCR Platforms
| Operational Parameter | Nanoplate dPCR | Droplet ddPCR |
|---|---|---|
| Workflow Integration | Fully integrated system [6] | Multiple instruments often required [6] |
| Time to Results | ~2 hours [6] | 6-8 hours [16] |
| Hands-on Time | Minimal [41] | Significant [6] |
| Throughput (samples/run) | Up to 1,248 (96-well plate) [6] | Up to 480 (5 plates) [6] |
| Sample Transfer Steps | Minimal (single plate) [16] | Multiple (tube to cartridge to plate) [6] |
| Contamination Risk | Lower [6] | Higher due to multiple transfers [6] |
| GMP Compliance Features | Available (21 CFR Part 11) [16] | Available [16] |
The streamlined workflow of ndPCR offers distinct advantages for quality control settings. As noted by RoslinCT, a contract development and manufacturing organization (CDMO), "For critical QC release assays in cell and gene therapy manufacturing, factors such as convenience, robustness, and streamlined workflows are paramount" [16]. The integrated "sample-in, results-out" process reduces hands-on time and minimizes opportunities for human error, which is crucial for release testing in regulated environments [16].
The workflow diagram above illustrates the fundamental operational differences between the two platforms. Nanoplate dPCR consolidates multiple steps into a single integrated instrument, while droplet ddPCR requires multiple discrete instruments and manual transfer steps, increasing complexity and hands-on time [6] [16].
Key Applications in Cell and Gene Therapy
Vector Copy Number (VCN) Quantification
Determining the number of vector genomes integrated into a host cell's genome is crucial for assessing the potency and safety of gene therapies. dPCR provides absolute quantification without reference standards, enabling more precise VCN calculation than qPCR methods [41]. This precision is vital given that viral transduction can be variable, and VCN serves as a critical metric for monitoring sustained transgene expression and determining therapeutic dose [41].
Both ndPCR and ddPCR platforms have demonstrated capability for VCN analysis, with studies showing high correlation between platforms when measuring the same samples [8]. However, the higher throughput and multiplexing capabilities of ndPCR platforms provide practical advantages for processing multiple patient samples simultaneously and measuring multiple critical quality attributes in a single run [16].
Quality Control Release Assays
Beyond VCN quantification, dPCR platforms support multiple essential QC functions in cell and gene therapy manufacturing:
- Virus Quantification: Precisely quantitating genomic titer, viral particle titer, and full/empty capsid ratios during purification and lot release to ensure accurate dosing [41]
- Purity & Safety Testing: Identifying product and process-related impurities such as residual host cell DNA, plasmid contaminants, or microbial contamination for in-process and release testing [41]
- Residual DNA Detection: Quantifying residual plasmid DNA post-electroporation in transfected cells [16]
- Genome Edit Detection: Absolute quantification of genome editing events generated by CRISPR-Cas9 or other genome editing tools [16]
For these applications, the consistency and reproducibility offered by dPCR platforms are essential. As noted in a quality control study, "Unlike qPCR, the number of AAV vector genomes determined by ddPCR was not affected by the primers and probes used, and no reference standards were required" [42]. This independence from external standards reduces a source of variability and makes the process more robust and efficient [41].
Experimental Protocols and Reagent Solutions
Typical Experimental Protocol for VCN Quantification
The following protocol summarizes key steps for vector copy number analysis using digital PCR platforms:
Sample Preparation: Extract genomic DNA from transduced cells using validated methods. For some targets, digest DNA with restriction enzymes (e.g., HaeIII or EcoRI) to improve accessibility, particularly for targets in tandem repeats [8]
Reaction Setup: Prepare dPCR master mix containing:
- DNA template (typically 1-100 ng)
- Primers and probes targeting the vector sequence
- Reference assay (e.g., for single-copy host gene)
- dPCR supermix
Optimization Note: Empirically adjust primer and probe concentrations within the manufacturer's validated framework to ensure optimal performance [3]
Partitioning and Amplification:
Data Analysis: Use platform-specific software to calculate absolute copy numbers based on Poisson statistics. Normalize vector copies to reference gene copies to determine VCN [41]
Essential Research Reagent Solutions
Table 3: Key Reagents for dPCR Assays in Cell and Gene Therapy
| Reagent Type | Specific Examples | Function & Application |
|---|---|---|
| Viral Titer Assays | Absolute Q Viral Titer Assays (eGFP, SV40 polyA, AAV ITR-2, CMV promoter) [41] | Quantification of viral vectors for gene therapy |
| Residual DNA Testing | resDNASEQ dPCR E1A DNA Fragment Length Kit; resDNASEQ dPCR E. coli DNA Kit [41] | Detect process-related impurities in final products |
| Lentivirus Characterization | ViralSEQ Lentivirus Physical Titer Kit; ViralSEQ Lentivirus Proviral DNA Titer Kit [41] | Quantitate lentiviral particles and measure vector integration |
| Restriction Enzymes | HaeIII, EcoRI [8] | Improve DNA accessibility for targets in tandem repeats |
| Custom Assay Design | Custom dPCR assays with self-service tools or design services [41] | Target-specific sequences not covered by pre-validated assays |
Platform Selection Guidelines
Decision Framework
Choosing between nanoplate and droplet dPCR technologies requires careful consideration of the specific application context:
For high-throughput QC release testing in GMP environments, ndPCR's integrated workflow, reduced hands-on time, and compliance features make it particularly suitable [16]
For research and process development where maximum sensitivity is required and workflow complexity is less critical, ddPCR's extremely low LOD may be advantageous [8]
For laboratories requiring multiplexing capability, ndPCR's ability to detect up to 5 targets simultaneously provides efficiency for complex assays [6]
For environments with budget constraints, total cost of ownership should be evaluated, including reagents, maintenance, and personnel time [6]
Regulatory Considerations
From a regulatory perspective, both technologies can be validated for use in clinical manufacturing under Good Manufacturing Practice (GMP) regulations [16]. Bio-Rad's ddPCR systems have established regulatory precedence, while emerging ndPCR platforms are rapidly catching up with features like 21 CFR Part 11 compliant software, Installation Qualification/Operational Qualification services, and comprehensive validation support [16]. The choice should be guided by specific assay requirements and the product's regulatory pathway, with demonstrated specificity, linearity, accuracy, and robustness being paramount regardless of platform [16].
Both nanoplate and droplet digital PCR platforms offer robust solutions for critical quality control applications in cell and gene therapy. While they demonstrate comparable performance in accuracy and precision for applications like vector copy number quantification, their operational characteristics differ significantly [8].
Nanoplate dPCR systems provide streamlined, integrated workflows better suited to the efficiency and compliance demands of GMP manufacturing environments [16]. Droplet dPCR systems offer exceptional sensitivity and have established regulatory precedence, though with more complex operational requirements [6] [42].
The decision between platforms should be guided by a fit-for-purpose evaluation considering throughput requirements, multiplexing needs, personnel resources, and regulatory strategy. As the field of advanced therapies continues to evolve, both technologies will play crucial roles in ensuring the safety and efficacy of these transformative medicines through precise molecular characterization.
Maximizing Performance: Troubleshooting Common Pitfalls and Optimization Strategies
Digital PCR (dPCR) has emerged as a powerful tool for the absolute quantification of nucleic acids, offering superior sensitivity and precision for applications ranging from clinical diagnostics to environmental monitoring. This guide provides an objective comparison of the performance characteristics between two main dPCR platforms: droplet digital PCR (ddPCR) and nanoplate digital PCR (ndPCR). Focusing on Limits of Detection (LOD) and Quantification (LOQ), we synthesize experimental data from recent studies to evaluate the sensitivity, accuracy, and precision of the Bio-Rad QX200 (ddPCR) and QIAGEN QIAcuity One (ndPCR) systems. The findings demonstrate that while both platforms deliver high performance, key differences in their operational parameters and susceptibility to experimental variables can guide researchers in selecting the optimal technology for their specific application needs.
Digital PCR (dPCR) represents the third generation of PCR technology, enabling absolute quantification of nucleic acids without the need for a standard curve [2]. The core principle involves partitioning a PCR reaction into thousands of individual reactions, so that each partition contains either zero, one, or a few nucleic acid molecules. Following end-point amplification, the fraction of positive partitions is counted, and the absolute concentration of the target sequence is calculated using Poisson statistics [28] [2]. This partitioning step is what gives dPCR its high sensitivity and precision, particularly at low target concentrations.
Two major partitioning methodologies have been commercially established:
- Droplet Digital PCR (ddPCR): This system, exemplified by the Bio-Rad QX200, uses a water-in-oil emulsion technology to partition samples into thousands of nanoliter-sized droplets [28] [2].
- Nanoplate Digital PCR (ndPCR): This system, such as the QIAGEN QIAcuity One, utilizes microfluidic chips containing arrays of nanoscale chambers to partition the reaction mixture [28] [43] [8].
Both platforms aim to achieve highly accurate and precise copy number estimations, but they rely on different mechanical and optical systems for partitioning and fluorescence reading, which can influence their performance in terms of LOD and LOQ.
Direct Comparison of LOD and LOQ
Quantitative Performance Metrics
The Limit of Detection (LOD) is the lowest concentration of an analyte that can be reliably distinguished from zero, while the Limit of Quantification (LOQ) is the lowest concentration that can be measured with acceptable precision and accuracy [28] [43] [27]. These parameters are critical for determining the suitability of a platform for applications requiring high sensitivity, such as detection of rare genetic mutations or low-abundance pathogens.
A direct comparative study using synthetic oligonucleotides and DNA from the ciliate Paramecium tetraurelia provided the following performance data for the two platforms [28] [8]:
Table 1: Direct comparison of LOD and LOQ between ndPCR and ddPCR platforms
| Platform | Partitioning Method | LOD (copies/µL input) | LOQ (copies/µL input) | Dynamic Range |
|---|---|---|---|---|
| QIAcuity One (ndPCR) | Nanoplate | 0.39 | 1.35 | <0.5 to >3000 copies/µL |
| QX200 (ddPCR) | Droplet | 0.17 | 4.26 | <0.5 to >3000 copies/µL |
This data reveals a trade-off between the two systems: the QX200 ddPCR demonstrated a slightly superior (lower) LOD, while the QIAcuity One ndPCR achieved a superior (lower) LOQ. Both platforms exhibited a wide dynamic range, with interpretable results from less than 0.5 copies/µL to over 3000 copies/µL input [28] [8].
Precision and Accuracy
Precision, often reported as the Coefficient of Variation (CV%), is a key metric for evaluating platform performance:
- For synthetic oligonucleotides, both platforms showed high precision above their LOQ thresholds, with CVs ranging between 6-13% for ddPCR and 7-11% for ndPCR [28] [8].
- With biological samples (e.g., DNA from P. tetraurelia), precision was notably influenced by the choice of restriction enzyme. For the QX200 ddPCR, using HaeIII instead of EcoRI significantly improved precision, reducing all CVs to below 5%. The QIAcuity One ndPCR showed less variability due to enzyme choice [28] [8].
- In copy number variation (CNV) studies, ddPCR demonstrated 95% concordance with the gold-standard pulsed field gel electrophoresis (PFGE) method, outperforming quantitative PCR (qPCR), which showed only 60% concordance [26].
Accuracy, determined by comparing expected versus measured gene copy numbers, was high for both dPCR platforms, with adjusted R² values of 0.98 for ndPCR and 0.99 for ddPCR [28] [8].
Experimental Protocols for Performance Validation
Protocol for LOD/LOQ Determination
The following methodology, adapted from cross-platform comparison studies, outlines a standardized approach for determining LOD and LOQ [28] [43] [8]:
Sample Preparation:
- Prepare a series of dilutions from a standardized DNA template (e.g., synthetic oligonucleotides or genomic DNA from a reference strain). The dilution series should span a wide concentration range, from expected single-copy levels to saturation.
- Accurately measure DNA concentrations using a fluorometer (e.g., Qubit Fluorometer) to ensure reliability, as concentrations provided by manufacturers may show deviations [28] [43].
dPCR Setup:
- ndPCR Protocol (QIAcuity One):
- Prepare a PCR mix compatible with the system (e.g., QIAcuity Probe PCR Master Mix).
- Load the mix and sample into a designated nanoplate (e.g., QIAcuity Nanoplate 26k/26k 24-Well). A typical reaction volume is 40 µL [28].
- Seal the plate and place it into the QIAcuity One instrument for partitioning, amplification, and imaging.
- ddPCR Protocol (QX200):
- Prepare a PCR mix using ddPCR Supermix for Probes.
- Load the mix and sample into a DG8 cartridge alongside droplet generation oil.
- Generate droplets using the QX200 Droplet Generator. A typical reaction volume is 20 µL, generating ~20,000 droplets [28] [39].
- Transfer the emulsified sample to a 96-well plate, seal, and perform PCR amplification in a thermal cycler.
- Read the plate using the QX200 Droplet Reader.
- ndPCR Protocol (QIAcuity One):
Data Analysis:
- Analyze data using the manufacturer's software (Quantasoft for QX200, QIAcuity Software Suite for QIAcuity One).
- LOD Calculation: Determine the lowest concentration where the target is reliably detected in all replicates. Statistical modeling (e.g., probit analysis) is often used [28] [27].
- LOQ Calculation: Determine the lowest concentration measurable with acceptable precision (typically CV < 25-35%). This can be established using a polynomial model fit to precision data across the dilution series [28] [43].
Figure 1: Experimental workflow for determining the Limit of Detection (LOD) and Limit of Quantification (LOQ) on nanoplate and droplet digital PCR platforms.
Protocol for Assessing Precision with Restriction Enzymes
The precision of gene copy number quantification, especially for organisms with high copy numbers or tandem repeats, can be influenced by DNA accessibility. Testing different restriction enzymes is a critical step for protocol optimization [28] [8]:
DNA Digestion:
- Select two or more restriction enzymes with different recognition sites and cutting frequencies (e.g., EcoRI and HaeIII).
- Perform enzymatic digestion on the sample DNA according to the manufacturer's specifications.
- Include an undigested control.
dPCR Analysis:
- Run the digested and undigested DNA samples in replicates (at least n=5) on both dPCR platforms.
- Use consistent PCR conditions and reagent concentrations across all samples.
Precision Calculation:
- For each sample and enzyme condition, calculate the mean copy number and standard deviation.
- Determine the Coefficient of Variation (CV%) as (Standard Deviation / Mean) × 100.
- Compare CV% across different restriction enzymes to identify the one that yields the highest precision (lowest CV%).
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of dPCR assays requires careful selection of reagents and materials. The following table lists key solutions used in the featured experiments.
Table 2: Key research reagent solutions for digital PCR experiments
| Reagent / Material | Function / Description | Example Use Case |
|---|---|---|
| Restriction Enzymes (e.g., HaeIII, EcoRI) | Digests genomic DNA to improve accessibility of target genes, enhancing precision [28] [8]. | Quantification of high-copy-number genes in ciliates [28] [8]. |
| Synthetic Oligonucleotides | Defined copy number standards for accurate calibration of LOD, LOQ, and dynamic range [28] [8]. | Platform performance validation and cross-platform comparisons [28]. |
| Fluorometer (e.g., Qubit) | Accurately measures DNA concentration of stock solutions, critical for calculating expected copy numbers [43] [39]. | Standardization of HAdV DNA for dPCR assay development [43]. |
| dPCR Supermix for Probes | Optimized reaction mix containing DNA polymerase, dNTPs, and buffer, tailored for probe-based detection in respective platforms [28] [39]. | Absolute quantification of Salmonella spp. using ddPCR [39]. |
| Target-specific Probes (e.g., FAM, HEX) | Fluorescently labeled hydrolysis probes (TaqMan) that provide sequence-specific detection in multiplex assays [43] [27]. | Detection of SARS-CoV-2 E gene and human adenovirus [43] [27]. |
Factors Influencing Platform Performance and Selection
Key Considerations for Platform Choice
The choice between ndPCR and ddPCR involves several technical and practical considerations that impact sensitivity, precision, and workflow efficiency.
Table 3: Key factors influencing performance and selection between dPCR platforms
| Factor | Impact on LOD/LOQ and Precision | Platform Comparison |
|---|---|---|
| Partitioning Technology | Higher number of partitions can improve dynamic range and precision. | ndPCR: Fixed number of partitions (e.g., 26,000 nanowell plate). ddPCR: Typically generates ~20,000 droplets per reaction [28] [2]. |
| Reaction Volume | Larger input volumes can enhance detection of very low concentration targets. | ndPCR: Typically uses 40 µL reaction volume. ddPCR: Typically uses 20 µL reaction volume [28]. |
| Restriction Enzyme Selection | Critical for precision when quantifying targets in complex genomic DNA, especially with high copy numbers or tandem repeats. | ddPCR precision improved significantly with HaeIII vs. EcoRI (all CVs <5%). ndPCR less affected by enzyme choice [28] [8]. |
| Workflow and Throughput | Affects practicality for high-throughput labs and risk of contamination. | ndPCR: Integrated, closed-system partitioning minimizes contamination and hands-on time [43]. ddPCR: Requires droplet generation and transfer steps, increasing hands-on time [2]. |
| Inhibition Resistance | Important for analysis of complex samples like wastewater or food. | Both platforms are more resistant to inhibitors than qPCR. ddPCR has demonstrated high tolerance to inhibitors in food and environmental samples [27] [37] [39]. |
Figure 2: Logical relationships between key experimental factors and digital PCR performance metrics. Factors like partitioning technology and reaction volume most directly influence LOD, while restriction enzyme choice and sample type are critical for precision.
Application-Specific Performance
- Clinical Diagnostics: Both platforms have proven effective for detecting low viral loads. A study on human adenovirus (HAdV) using ndPCR achieved an LOD and LOQ of 0.95 cp/μL, highlighting its utility for monitoring immunocompromised patients [43].
- Food Safety and Environmental Monitoring: ddPCR has demonstrated superior sensitivity compared to real-time PCR for detecting Lacticaseibacillus casei in milk, with a limit of detection of 100 CFU/mL [37]. Similarly, for SARS-CoV-2 in wastewater, both RT-ddPCR and RT-qPCR showed similar performance during low-incidence periods, suggesting that platform choice may depend on available resources and throughput requirements rather than sensitivity alone [27].
- Allergen Detection: ndPCR has shown improved sensitivity over real-time PCR for tracing allergenic foods like sesame, detecting levels as low as 0.1 mg/kg in processed biscuits [22].
The comparative analysis of ndPCR and ddPCR platforms reveals that both technologies offer high sensitivity, precision, and absolute quantification for nucleic acid detection. The QIAcuity One (ndPCR) demonstrated a slightly better LOQ, while the QX200 (ddPCR) showed a marginally superior LOD. The choice between platforms should be guided by specific application needs: ndPCR offers a streamlined, integrated workflow advantageous for routine high-throughput settings, whereas ddPCR provides proven robustness for complex samples and flexibility in assay design. Factors such as restriction enzyme optimization are critical for maximizing precision, especially for challenging targets. Ultimately, both platforms represent powerful tools for advancing research and diagnostics where precise nucleic acid quantification is paramount.
This guide provides an objective comparison of the precision and accuracy of nanoplate-based digital PCR (dPCR) and droplet digital PCR (ddPCR). The evaluation, grounded in empirical data, focuses on key performance metrics such as the Coefficient of Variation (CV) and concordance with reference methods. While both platforms demonstrate the high precision and accuracy characteristic of digital PCR, their performance can vary based on specific experimental conditions, such as the use of restriction enzymes and the target copy number. Nanoplate-based systems often show operational advantages in workflow simplicity and reduced contamination risk, whereas ddPCR systems boast a long track record of use and extensive validation data. The choice between platforms should be guided by the specific application, required throughput, and the need for multiplexing.
Digital PCR (dPCR) is a powerful technique for the absolute quantification of nucleic acids. Its principle involves partitioning a PCR reaction into thousands of individual reactions, each yielding a binary result (positive or negative for the target). The absolute copy number concentration is then calculated using Poisson statistics [44]. This method eliminates the need for a standard curve, a potential source of inaccuracy in quantitative PCR (qPCR).
In evaluating dPCR performance, two distinct yet related metrics are paramount:
- Accuracy refers to the closeness of agreement between a measured value and its true or accepted reference value [44]. It is often assessed through concordance studies with a gold-standard method.
- Precision describes the variability in independent measurement results obtained from the same sample under stipulated conditions [44]. It is typically reported as the Coefficient of Variation (CV), which is the ratio of the standard deviation to the mean, expressed as a percentage. A lower CV indicates higher precision.
This guide interprets these metrics to compare the performance of nanoplate-based dPCR and droplet-based ddPCR platforms.
Quantitative Performance Data Comparison
The following tables consolidate key experimental data from recent studies to facilitate a direct comparison of the two platforms.
Table 1: Comparative Limits of Detection and Quantification
| Platform | Specific System | Limit of Detection (LOD) | Limit of Quantification (LOQ) | Application Context | Source |
|---|---|---|---|---|---|
| Nanoplate dPCR | QIAcuity One | 0.39 copies/µL [8] | 1.35 copies/µL [8] | Copy number analysis in protists [8] | |
| Droplet ddPCR | QX200 | 0.17 copies/µL [8] | 4.26 copies/µL [8] | Copy number analysis in protists [8] | |
| Nanoplate dPCR | QIAcuity | 0.9476 cp/µL [43] | 0.9476 cp/µL [43] | Human adenovirus detection [43] | |
| Nanoplate RT-dPCR | QIAcuity | 1.83 copies/µL [45] | Not specified | Canine respiratory coronavirus [45] |
Table 2: Precision and Accuracy Data from Platform Comparison Studies
| Platform | Precision (CV) with Synthetic Oligos | Precision (CV) with Biological DNA (EcoRI) | Precision (CV) with Biological DNA (HaeIII) | Accuracy (Concordance with Reference) | Application Context |
|---|---|---|---|---|---|
| Nanoplate dPCR | 7 - 11% [8] | 0.6% - 27.7% [8] | 1.6% - 14.6% [8] | High linearity (R²adj=0.98) with oligos [8] | Gene copy number in Paramecium [8] |
| Droplet ddPCR | 6 - 13% [8] | 2.5% - 62.1% [8] | < 5% (all samples) [8] | 95% concordance with PFGE [26] | DEFA1A3 CNV in human genomics [26] |
Table 3: Workflow and Practical Considerations
| Parameter | Nanoplate-based dPCR | Droplet-based ddPCR |
|---|---|---|
| Partitioning Mechanism | Fixed wells/nanochannels on a plate [19] [16] | Water-in-oil emulsion droplets [16] |
| Typical Workflow | Integrated, "sample-to-result" automated system [16] | Multiple steps and instruments; requires droplet generation and transfer [16] |
| Hands-on Time | Shorter, streamlined [16] | Longer, more manual steps [16] |
| Risk of Contamination | Lower (closed system, fewer transfers) [19] [16] | Higher (during droplet transfer) [19] |
| Multiplexing Capability | Available for 4-12 targets [16] | Limited in older models, newer ones can detect up to 12 targets [16] |
| Ideal Environment | QC and regulated environments [16] | Research and development labs [16] |
Experimental Protocols for Key Comparisons
Protocol: Cross-Platform Precision and Accuracy Assessment
This protocol is derived from a study directly comparing the QIAcuity One (nanoplate) and QX200 (droplet) systems [8].
1. Sample Preparation:
- Synthetic Standards: Use a dilution series of synthetic oligonucleotides with known concentrations to establish a dynamic range (e.g., from <0.5 to >3000 copies/µL).
- Biological DNA: Extract DNA from a model organism (e.g., the ciliate Paramecium tetraurelia) with varying cell counts. Treat DNA samples with different restriction enzymes (e.g., EcoRI and HaeIII) to assess their impact on precision.
2. dPCR Reaction Setup:
- Nanoplate dPCR: Prepare a PCR mix and load it directly into a QIAcuity nanoplate. The instrument performs partitioning, thermocycling, and imaging automatically [45].
- Droplet ddPCR: Prepare a PCR mix and use a droplet generator to create an oil-water emulsion containing ~20,000 droplets. Transfer the droplets to a PCR plate for thermocycling, then load the plate into a droplet reader for analysis [44].
3. Data Analysis:
- Limit of Detection/Quantification (LOD/LOQ): Analyze the dilution series data. LOD is the lowest concentration distinguishable from zero. LOQ is the lowest concentration measurable with acceptable precision (CV < 35% is common), determined via model fitting (e.g., 3rd degree polynomial) [8].
- Precision: For each sample type and condition, run multiple technical replicates. Calculate the mean, standard deviation, and Coefficient of Variation (CV) for the measured copy number.
- Accuracy: For synthetic standards, compare measured copies/µL to the expected value. For biological samples, assess linearity (R²) across cell number dilutions and/or concordance with a gold-standard method like Pulsed Field Gel Electrophoresis (PFGE) [26].
Protocol: Validation of a ddPCR Assay for Copy Number Variation
This protocol outlines the key steps for validating ddPCR accuracy against a gold standard, as performed in a human genomics study [26].
- 1. Sample Selection: Use genomic DNA samples with copy number variants (CNVs) previously characterized by a reference method (e.g., PFGE for the DEFA1A3 locus). Include samples covering the entire CNV range (e.g., 2 to 12 copies).
- 2. Assay Design: Design and optimize TaqMan assays targeting the CNV region and a reference gene (assumed to be two copies per diploid genome).
- 3. Parallel Testing: Run all samples on both the ddPCR platform and the reference PFGE method.
- 4. Concordance Analysis: Classify results as concordant if the ddPCR and PFGE copy numbers are within ±1 copy. Calculate the overall concordance rate and perform statistical analyses (e.g., Spearman correlation, linear regression).
Interpreting CV and Concordance Data
Understanding Coefficient of Variation (CV) Trends
The data in Table 2 reveals several critical trends. Both platforms show good and comparable precision (CVs of 6-13%) with clean synthetic templates [8]. However, with complex biological DNA, precision can be significantly impacted by factors such as the choice of restriction enzyme. In one study, ddPCR's precision improved dramatically (CVs from >60% to <5%) when using HaeIII instead of EcoRI, suggesting enzyme choice affects template accessibility, particularly in systems with tandemly repeated genes [8]. Nanoplate dPCR also showed improved precision with HaeIII, but the effect was less pronounced, indicating platform-specific differences in handling complex DNA [8].
Assessing Accuracy Through Concordance
Concordance analysis measures the agreement between a new method and an established gold standard. A high concordance rate (e.g., 95% between ddPCR and PFGE [26]) provides strong evidence of the new method's accuracy. In contrast, qPCR showed only 60% concordance with PFGE in the same study, highlighting dPCR's superior accuracy, especially at higher copy numbers where qPCR's fold-ratio relationship breaks down [26]. Furthermore, nanoplate dPCR has demonstrated high accuracy in detecting and quantifying targets in complex matrices like processed meat, showing 100% concordance with product labels, a performance that surpassed that of real-time PCR [19].
The Scientist's Toolkit: Essential Reagents and Materials
Table 4: Key Research Reagent Solutions for dPCR Experiments
| Item | Function | Example Use-Case |
|---|---|---|
| Restriction Enzymes | Digests long DNA strands to ensure free access of target sequences and prevent bias from linked copies. Critical for precision in copy number analysis. | HaeIII was found to significantly improve precision compared to EcoRI in a protist gene copy number study [8]. |
| Synthetic Oligonucleotides | Provide a known, pure standard for establishing calibration curves, determining LOD/LOQ, and assessing accuracy without sample matrix effects. | Used to evaluate the dynamic range, accuracy, and precision of both nanoplate and droplet systems [8]. |
| Certified Reference Materials (CRMs) | Materials with a certified copy number concentration, used for method validation and ensuring measurement traceability. | ERM-AD623 plasmid solutions were used for the in-house validation of a ddPCR method [44]. |
| dPCR Supermix | Optimized buffer containing DNA polymerase, dNTPs, and MgCl₂, formulated for efficient amplification in partitioned reactions. | A core component of the PCR mix in both Bio-Rad's ddPCR [44] and QIAGEN's ndPCR systems. |
| Fluorophore-Labeled Probes | Sequence-specific probes (e.g., TaqMan) that generate a fluorescent signal upon amplification, enabling target detection and multiplexing. | Used for specific detection of meat species (pork, chicken) in a duplex nanoplate dPCR assay [19]. |
The decision between nanoplate dPCR and droplet ddPCR should be driven by the specific application and operational needs. The experimental data shows that both platforms are capable of high precision and accuracy, but their performance can be influenced by sample type and preparation.
- Choose Nanoplate dPCR if: Your priority is a streamlined, automated workflow with a lower risk of contamination, making it ideal for quality control (QC) environments and clinical diagnostics [45] [16]. Its consistent partitioning and robust performance with complex samples [8] [19] are significant advantages.
- Choose Droplet ddPCR if: You require a platform with an extensive history of peer-reviewed validation data, particularly for research applications. It demonstrates excellent accuracy and precision, as validated against gold-standard methods like PFGE [26].
For all applications, careful assay optimization—including the critical choice of restriction enzymes—is essential for achieving the highest levels of precision and accuracy with either platform.
The Impact of Restriction Enzymes and Assay Design on Precision
In the evolving landscape of molecular diagnostics and research, digital PCR (dPCR) has emerged as a powerful tool for absolute nucleic acid quantification, offering superior precision and sensitivity compared to traditional quantitative PCR (qPCR). This technology has become particularly valuable in diverse fields, including environmental monitoring, clinical virology, and cell and gene therapy development [8] [3]. The core principle of dPCR involves partitioning a sample into thousands of individual reactions, enabling binary detection (positive or negative) of target molecules and absolute quantification through Poisson statistics [8].
Two major dPCR platform types have gained widespread adoption: droplet digital PCR (ddPCR), which uses a water-oil emulsion to create nanoliter-sized droplets, and nanoplate-based dPCR (ndPCR), which distributes samples across fixed micro-wells [16]. While these platforms share a common underlying principle, their partitioning mechanisms, workflows, and susceptibility to experimental variables differ significantly. Among the critical factors influencing measurement precision is the strategic use of restriction enzymes during assay design, which can enhance data accuracy by improving template accessibility, particularly for complex or repetitive genomic targets [8] [46].
This guide objectively compares the performance of nanoplate and droplet digital PCR systems, focusing specifically on how restriction enzyme selection and overall assay design impact result precision. By synthesizing current experimental data, we provide researchers and drug development professionals with evidence-based insights to optimize their dPCR applications.
Comparative Platform Performance and Workflow
Key Technological Differences
The fundamental difference between nanoplate and droplet dPCR systems lies in their partitioning mechanisms. Nanoplate-based systems (e.g., QIAGEN QIAcuity) utilize microfluidic chips containing fixed nanowells, typically numbering around 20,000-26,000, to partition samples [3] [16]. This creates a closed system that minimizes contamination risk and streamlines workflow. In contrast, droplet-based systems (e.g., Bio-Rad QX200) generate 20,000 or more nanoliter-sized water-in-oil droplets through emulsion processes, requiring multiple instruments and manual transfer steps [8] [16].
These mechanistic differences translate to distinct practical implications. Nanoplate systems offer a "sample-in, results-out" process completed in approximately 90 minutes, significantly faster than the 6-8 hours typically required for ddPCR workflows [16]. The integrated nature of nanoplate systems reduces hands-on time and potential for human error, making them particularly suitable for quality control environments where reproducibility and efficiency are paramount [16].
Workflow Visualization
The following diagram illustrates the key procedural differences between nanoplate and droplet digital PCR workflows, highlighting steps where restriction enzymes may be incorporated and where variability can influence precision:
Performance Comparison Data
Direct comparisons of nanoplate and droplet dPCR platforms reveal important performance differences. A 2025 study systematically evaluated the QX200 ddPCR system and QIAcuity One ndPCR system using synthetic oligonucleotides and DNA from the ciliate Paramecium tetraurelia, providing quantitative data on sensitivity and precision [8].
Table 1: Comparison of Sensitivity Metrics Between dPCR Platforms
| Performance Metric | Nanoplate dPCR (QIAcuity) | Droplet dPCR (QX200) |
|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL input (15.60 copies/reaction) | 0.17 copies/µL input (3.31 copies/reaction) |
| Limit of Quantification (LOQ) | 1.35 copies/µL input (54 copies/reaction) | 4.26 copies/µL input (85.2 copies/reaction) |
| Dynamic Range | 0.9476 to 770.4 cp/µL (for HAdV assay) [43] | Optimized for 20,000 copies/µL input [47] |
| Best Model Fit for LOQ | 3rd degree polynomial [8] | 3rd degree polynomial [8] |
The data demonstrates a trade-off between detection and quantification capabilities. While the ddPCR system showed a slightly lower LOD, the ndPCR platform achieved a lower LOQ, indicating better performance for reliable quantification at very low target concentrations [8]. Both platforms showed high correlation between expected and measured gene copy numbers (ndPCR: R²adj = 0.98; ddPCR: R²adj = 0.99), though measured values were consistently lower than expected for both systems [8].
Impact of Restriction Enzymes on Precision
Restriction Enzymes as Precision Modulators
Restriction enzymes play a crucial role in enhancing dPCR precision by digesting DNA templates to improve accessibility of target regions, particularly for complex genomic DNA or templates with secondary structures. Their application has demonstrated significant platform-dependent effects on measurement variability [8].
A comprehensive study comparing ddPCR and ndPCR performance found that restriction enzyme selection directly impacted precision, with effects more pronounced in droplet-based systems. When analyzing DNA from Paramecium tetraurelia, the choice between EcoRI and HaeIII restriction enzymes resulted in markedly different precision profiles [8].
Table 2: Effect of Restriction Enzymes on Precision Across dPCR Platforms
| Experimental Condition | Platform | Precision (Average %CV) | Notes |
|---|---|---|---|
| EcoRI digestion | ddPCR (QX200) | 2.5% - 62.1% | High variability, especially at lower cell counts |
| HaeIII digestion | ddPCR (QX200) | < 5% | Consistently low CV across all cell numbers |
| EcoRI digestion | ndPCR (QIAcuity) | 0.6% - 27.7% | Moderate variability, better than ddPCR with EcoRI |
| HaeIII digestion | ndPCR (QIAcuity) | 1.6% - 14.6% | Good precision, less dramatic improvement than ddPCR |
The data reveals that HaeIII digestion dramatically improved precision for ddPCR, reducing maximum CV values from over 60% to below 5%. This suggests that HaeIII more effectively digested the target DNA, leading to more consistent partitioning and amplification [8]. The ndPCR system showed less sensitivity to restriction enzyme choice, indicating potentially more robust performance across different assay conditions.
Mechanisms of Restriction Enzyme Effects
The improvement in precision observed with specific restriction enzymes can be attributed to several factors. Restriction enzymes digest DNA at specific recognition sites, fragmenting long DNA molecules that might otherwise partition inefficiently or irregularly across reaction chambers [46]. This is particularly important for targets with tandem repeats or complex secondary structures, where accessibility varies significantly between digested and undigested templates [8].
HaeIII recognizes and cuts at GG▼CC sites, while EcoRI cuts at G▼AATTC sites. The different sequence specificity and cutting efficiency of these enzymes directly impact template accessibility during PCR amplification. The dramatic precision improvement observed with HaeIII in ddPCR suggests it may be particularly effective for preparing the specific genomic targets found in Paramecium tetraurelia, possibly due to more optimal distribution of restriction sites around the target region [8].
Experimental Protocols for Method Validation
Protocol 1: Assessing Platform Precision with Restriction Enzymes
The following protocol is adapted from the 2025 study comparing ddPCR and ndPCR platforms [8]:
Sample Preparation: Prepare DNA samples from a known number of Paramecium tetraurelia cells (e.g., 10, 50, 100 cells) using standard extraction methods. Alternatively, use synthetic oligonucleotides with known concentrations.
Restriction Enzyme Digestion: Set up separate digestion reactions for each sample using:
- 1µg DNA template
- 10U of restriction enzyme (EcoRI or HaeIII)
- Appropriate reaction buffer
- Incubate at 37°C for 30 minutes, followed by enzyme inactivation at 65°C for 20 minutes
dPCR Reaction Setup:
- Prepare master mixes according to manufacturer specifications for each platform
- For ndPCR: Use QIAcuity Probe PCR Kit and load 40µL reactions into nanoplate wells
- For ddPCR: Use ddPCR Supermix for Probes and generate droplets following manufacturer protocols
- Use identical primer-probe combinations for both platforms targeting the gene of interest
Amplification and Analysis:
- Run endpoint PCR with optimized cycling conditions
- Analyze partitions using platform-specific software
- Calculate copy numbers using Poisson statistics
Precision Assessment:
- Perform minimum of 3 technical replicates per condition
- Calculate coefficient of variation (%CV) for each sample set
- Compare precision profiles across platforms and restriction enzyme conditions
Protocol 2: Determining Limit of Detection and Quantification
This protocol, adapted from multiple validation studies [8] [43] [47], provides a standardized approach for establishing assay sensitivity:
Reference Material Preparation: Use synthetic oligonucleotides or plasmid DNA containing the target sequence. Precisely quantify using fluorometric methods (e.g., Qubit Fluorometer).
Sample Dilution Series: Prepare a minimum of 6 dilution levels covering the expected dynamic range, from below expected LOD to above expected LOQ. Include dilution levels that will likely oversaturate the system to establish upper limits.
Replicate Testing: Analyze each dilution level in at least 4 technical replicates across multiple independent runs to assess inter-run and intra-run variability.
Data Analysis:
- LOD Determination: Identify the lowest concentration where the target is detected in ≥95% of replicates
- LOQ Determination: Using the best model fit (e.g., 3rd degree polynomial), identify the lowest concentration where the CV is ≤25% [8] [47]
- Dynamic Range Establishment: Define the range where linearity is maintained (R² ≥ 0.98) and quantification remains accurate
Research Reagent Solutions
Successful implementation of precise dPCR assays requires careful selection of reagents and materials. The following table outlines key solutions and their functions based on the evaluated studies:
Table 3: Essential Research Reagents for dPCR Assay Development
| Reagent/Material | Function | Considerations for Precision |
|---|---|---|
| Restriction Enzymes (HaeIII, EcoRI) | Digest genomic DNA to improve template accessibility | Enzyme selection significantly impacts precision, especially for ddPCR [8] |
| dPCR Supermix | Provides optimized buffer, enzymes, and nucleotides for amplification | Master mix choice is critical for accurate copy number quantification [31] |
| Fluorophore-Labeled Probes | Target-specific detection using FAM, VIC, or HEX channels | Proper concentration optimization minimizes background and improves cluster separation [47] |
| Synthetic Oligonucleotides | Reference standards for quantification and validation | Must be accurately quantified using fluorometry, not spectrophotometry [8] |
| Nanoplate or Cartridge | Reaction vessel for partitioning | Fixed well systems minimize volume variation between partitions [16] |
| Droplet Generation Oil | Creates water-in-oil emulsion for partitioning | Stability of droplets affects result reliability; overnight cooling can improve statistical power [31] |
The selection between nanoplate and droplet digital PCR platforms involves careful consideration of precision requirements, workflow constraints, and specific application needs. Evidence from comparative studies indicates that while both platforms can deliver highly precise results, their performance is significantly influenced by assay design choices, particularly restriction enzyme selection.
Nanoplate systems offer streamlined, automated workflows advantageous for quality control environments, with demonstrated robustness across different restriction enzyme conditions. Droplet systems provide exceptional sensitivity for detection but show greater variability in precision that can be dramatically improved through optimal restriction enzyme selection.
The data consistently shows that HaeIII restriction enzyme digestion significantly enhances precision in droplet-based systems, reducing CV values from over 60% to below 5% in comparative studies [8]. This finding underscores the critical importance of matching not only platform selection but also enzymatic digestion strategies to specific sample types and targets.
For researchers requiring the utmost precision in nucleic acid quantification, particularly with challenging sample types or complex genetic targets, thorough validation of both platform performance and restriction enzyme efficacy is essential. The protocols and comparative data presented here provide a foundation for such method development, enabling informed decision-making for precise molecular quantification in research and diagnostic applications.
Digital PCR (dPCR) represents the third generation of PCR technology, enabling absolute quantification of nucleic acids without the need for standard curves [2]. This technique works by partitioning a PCR mixture into thousands of individual reactions, each containing zero, one, or a few target molecules [2]. Following end-point amplification, the fraction of positive partitions is counted, and the absolute target concentration is calculated using Poisson statistics [2]. The two primary dPCR platform architectures are droplet digital PCR (ddPCR), which uses water-in-oil emulsion droplets, and nanoplate digital PCR (ndPCR), which employs microchambers embedded in a solid chip [2]. Both platforms have demonstrated superior performance compared to quantitative PCR (qPCR) in applications requiring high sensitivity and precision [48].
A significant challenge in molecular diagnostics is the presence of PCR inhibitors in complex sample matrices. Formalin-fixed, paraffin-embedded (FFPE) tissues, a cornerstone of clinical pathology, contain cross-linking agents and fragmentation that impede amplification efficiency [5]. Similarly, food matrices comprise polysaccharides, phenolics, and fats that co-extract with DNA and inhibit polymerases [49] [50]. These inhibitors can skew quantification results, leading to false negatives or inaccurate measurements. This review objectively compares the robustness of nanoplate and droplet dPCR platforms when analyzing these challenging samples, providing researchers with experimental data to guide platform selection.
Performance Comparison in FFPE and Food Matrices
Quantitative Performance Metrics Across Platforms
Table 1: Comparison of dPCR Platform Performance in Complex Matrices
| Performance Metric | Nanoplate dPCR (QIAcuity) | Droplet Digital PCR (QX200) | Application Context |
|---|---|---|---|
| Correlation with ddPCR | r = 0.954 [5] | Benchmark | CDH13 methylation in FFPE breast cancer samples [5] |
| Sensitivity | 99.08% [5] | 98.03% [5] | CDH13 methylation detection [5] |
| Specificity | 99.62% [5] | 100% [5] | CDH13 methylation detection [5] |
| Tolerance to Inhibition | Less sensitive to inhibitors due to integrated partitioning [20] | Greater tolerance to inhibitors than qPCR [49] | GMO quantification in food/feed [49] [20] |
| Partition Number | ~26,000 partitions/well [20] | ~20,000 droplets/reaction [5] [49] | Standard configuration |
| Dynamic Range | Wide dynamic range demonstrated [51] | 4 orders of magnitude [49] | Absolute quantification |
| Limit of Detection (LOD) | Not explicitly stated | 5 target DNA copies [49] | GMO quantification |
| Throughput & Workflow | Integrated partitioning, thermocycling, and imaging [20] | Requires droplet generation and transfer steps [20] | Routine laboratory testing |
Analysis of Comparative Performance Data
Experimental data from direct platform comparisons reveal highly comparable analytical performance in absolute quantification. A 2025 study analyzing CDH13 gene methylation in 141 FFPE breast cancer tissues found an exceptionally strong correlation (r = 0.954) between nanoplate (QIAcuity) and droplet (QX200) platforms [5]. Both systems demonstrated similar sensitivity (99.08% vs. 98.03%) and specificity (99.62% vs. 100%) with no statistically significant differences [5].
The primary differentiators emerge in workflow efficiency and inhibitor tolerance. Nanoplate systems offer fully integrated workflows where partitioning, thermocycling, and imaging occur within a single instrument, reducing manual handling [20]. Droplet systems require separate instruments for droplet generation, thermocycling, and reading [20]. For inhibitor tolerance, both dPCR platforms outperform qPCR, with ndPCR showing particular robustness in GMO quantification in complex food matrices [49] [20].
Experimental Protocols for Assessing Platform Robustness
DNA Extraction and Quality Control from FFPE Samples
The following protocol, adapted from a CDH13 methylation study, ensures high-quality DNA extraction from challenging FFPE samples [5]:
- Deparaffinization and Lysis: Cut FFPE tissue sections (10µm thickness) and process using xylene for deparaffinization. Transfer deparaffinized tissue to a microcentrifuge tube and incubate with proteinase K at 56°C for tissue digestion [5] [52].
- DNA Extraction and Purification: Isolate genomic DNA using commercially available kits (e.g., DNeasy Blood and Tissue kit, Qiagen). For FFPE-derived DNA, additional purification steps may be necessary to remove formalin-induced crosslinks [5].
- DNA Quantification and Quality Assessment: Quantify DNA concentration using fluorescence-based methods (e.g., Qubit dsDNA BR Assay). Assess DNA quality via spectrophotometric ratios (A260/280 ~1.8-2.0) and fragment analysis [5] [52].
- Bisulfite Conversion (Methylation Analysis): For methylation studies, treat 1 µg of isolated DNA with bisulfite using commercial kits (e.g., EpiTect Bisulfite kit, Qiagen) according to manufacturer's instructions [5].
Digital PCR Assay Setup and Optimization
Table 2: Standardized Reaction Conditions for dPCR Platforms
| Reaction Component | Nanoplate dPCR (QIAcuity) | Droplet Digital PCR (QX200) |
|---|---|---|
| Reaction Volume | 12-40 µL [5] [20] | 20-22 µL [5] [49] |
| Master Mix | QIAcuity 4× Probe PCR Master Mix [5] | Supermix for Probes (No dUTP) [5] |
| Primer Concentration | 0.96 µL of forward/reverse primer (10 µM stock) [5] | 0.45 µL of forward/reverse primer (10 µM stock) [5] |
| Probe Concentration | 0.48 µL of each probe (10 µM stock) [5] | 0.45 µL of each probe (10 µM stock) [5] |
| DNA Template | 2.5 µL [5] | 2.5 µL [5] |
| Partitioning Method | Automated nanoplate partitioning [20] | Droplet generation oil for probes [5] |
| Thermal Cycling | Initial activation: 95°C for 2 min; 40 cycles: 95°C for 15s, 57°C for 1min [5] | Initial denaturation: 95°C for 10min; 40 cycles: 94°C for 30s, 57°C for 1min [5] |
Data Analysis and Validation
- Threshold Setting: Manually set fluorescence thresholds based on positive and negative controls, considering signal amplitude and binding specificity [5]. For the QIAcuity system, a threshold of 45 was optimal for CDH13 methylation analysis [5].
- Quality Control Metrics: Establish acceptance criteria for valid runs. In CDH13 methylation analysis, samples required >7,000 valid partitions and at least 100 positive partitions [5].
- Absolute Quantification: Calculate target concentration using Poisson statistics based on the fraction of positive and negative partitions [2]. For copy number variation analysis, calculate the ratio of target to reference gene copies [51] [52].
- Inhibition Testing: Perform serial dilutions of DNA samples. A sample is considered inhibited if the calculated concentration at higher dilutions differs by more than 25% from the concentrated sample [20].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for dPCR in Complex Matrices
| Reagent/Material | Function | Example Products |
|---|---|---|
| Nucleic Acid Extraction Kits | Isolation of high-quality DNA from complex matrices | DNeasy Blood & Tissue Kit (Qiagen) [5], NucleoSpin Food Kit [49], Maxwell RSC ccfDNA Plasma Kit [51] |
| Bisulfite Conversion Kits | DNA modification for methylation analysis | EpiTect Bisulfite Kit (Qiagen) [5] |
| dPCR Master Mixes | Optimized buffers/enzymes for partition-based amplification | QIAcuity Probe PCR Master Mix [5], ddPCR Supermix for Probes [5] |
| Reference Gene Assays | Endogenous controls for normalization and quality control | Lectin gene for soybean [20], multi-copy reference genes [51] |
| Partitioning Consumables | Generation of nanoscale reactions | QIAcuity Nanoplates [5] [20], DG8 Cartridges & Droplet Generation Oil [5] |
| Fluorophore-Labeled Probes | Target-specific detection | FAM, HEX/VIC-labeled TaqMan probes [5] [50] |
| Certified Reference Materials | Method validation and standardization | ERM-BF410 series (JRC) [20], AOCS reference materials [20] |
Workflow and Platform Selection Guidance
Key Selection Criteria
- Sample Throughput Requirements: Nanoplate systems offer streamlined processing of multiple samples in parallel with minimal manual intervention, making them ideal for clinical laboratories with high sample volumes [20].
- Inhibition Resistance Needs: For highly inhibitory matrices, nanoplate dPCR demonstrates robust performance due to consistent partition integrity and reduced handling variability [20].
- Multiplexing Complexity: Droplet systems provide established flexibility for complex multiplexing applications, though nanoplate systems are advancing in this capability [50].
- Existing Laboratory Infrastructure: Laboratories with extensive qPCR experience may find droplet systems more compatible with existing workflows and plasticware [49].
Both nanoplate and droplet digital PCR platforms demonstrate exceptional and comparable performance for nucleic acid quantification in challenging matrices like FFPE tissues and complex food samples. The strong correlation (r = 0.954) between platforms for DNA methylation analysis in FFPE tissues underscores their reliability [5]. The decision between platforms should be guided by specific application requirements: nanoplate dPCR offers advantages in workflow integration and inhibitor tolerance, while droplet dPCR provides established flexibility and a proven track record. As both technologies continue to evolve, their implementation will undoubtedly expand the frontiers of precision molecular analysis in complex sample matrices.
Optimizing Throughput and Multiplexing Capabilities for Complex Assays
Digital PCR (dPCR) represents a significant advancement in nucleic acid quantification by enabling absolute target measurement without the need for standard curves [2]. This technology partitions a PCR reaction into thousands of individual reactions, allowing precise counting of target molecules through Poisson statistical analysis [8] [28]. The two primary dPCR platforms—nanoplate-based digital PCR (ndPCR) and droplet digital PCR (ddPCR)—differ fundamentally in their partitioning mechanisms, which directly impacts their performance in throughput, multiplexing, and precision for complex assays [6] [16].
This comparison guide objectively evaluates ndPCR and ddPCR platforms within the broader context of precision and accuracy research. As dPCR gains prominence in diverse fields including oncology, microbiology, and cell and gene therapy, understanding the technical capabilities and limitations of each system becomes crucial for researchers, scientists, and drug development professionals seeking to optimize their analytical workflows [2] [16].
Technology Comparison: Partitioning Mechanisms and Workflows
Partitioning Approaches and Their Implications
The core differentiation between ndPCR and ddPCR lies in their partitioning methodologies, which directly influence workflow complexity, reproducibility, and data quality [6].
Nanoplate-based dPCR (ndPCR): This system utilizes microfluidic chips or nanoplates containing fixed arrays of microscopic wells. Partitions are created through capillary action or active fluidic control, typically generating 8,500-26,000 partitions per well with volumes of approximately 10 nL [6]. The process is fully integrated within a single instrument that performs partitioning, thermocycling, and imaging [30] [29].
Droplet-based dPCR (ddPCR): This approach employs water-oil emulsion technology to generate 20,000 to millions of partitions as nanoliter-sized droplets (10-100 pL) [6] [2]. The workflow requires multiple instruments including a droplet generator, thermocycler, and droplet reader [6].
The fundamental workflow differences between these technologies significantly impact their practical implementation in research settings, as illustrated below:
Comparative Platform Specifications
Different dPCR platforms offer varying capabilities in partition numbers, multiplexing capacity, and throughput. The table below summarizes key specifications for major commercial systems:
Table 1: Digital PCR Platform Comparison Specifications
| Platform | Type | Partitions per Well | Multiplexing Capacity | Throughput (8-hour shift) | Time to Result |
|---|---|---|---|---|---|
| QIAcuity One 5plex | Nanoplate | 8,500-26,000 [6] | Up to 12-plex [53] | 480 samples (96-well) [30] | ~2 hours [30] |
| QIAcuity Eight | Nanoplate | 8,500-26,000 [6] | Up to 12-plex [53] | 1,536 samples (96-well) [30] | First plate ~2 hours [30] |
| Bio-Rad QX200/QX600 | Droplet | ~20,000 [6] [16] | Up to 6-plex (QX600) [16] | Information missing | 6-8 hours [16] |
| Bio-Rad QX700 | Droplet | Information missing | Up to 12-plex [16] | Information missing | Information missing |
| Thermo Fisher Absolute Q | Chip-based | ~20,000 [16] | Information missing | Information missing | Information missing |
| Stilla Naica System | Microfluidic chip | ~20,000-30,000 [6] | Up to 3-plex [6] | 24 samples [6] | 2-3 hours [6] |
Performance Comparison: Experimental Data
Precision and Sensitivity Analysis
A 2025 study directly compared the QIAcuity ndPCR system (QIAGEN) with the QX200 ddPCR system (Bio-Rad) using synthetic oligonucleotides and DNA from Paramecium tetraurelia cells [8] [28]. The research examined limits of detection (LOD), limits of quantification (LOQ), and precision across both platforms.
Table 2: Sensitivity and Precision Performance Metrics
| Parameter | QIAcuity ndPCR | QX200 ddPCR |
|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/μL input [8] [28] | 0.17 copies/μL input [8] [28] |
| Limit of Quantification (LOQ) | 1.35 copies/μL input [8] [28] | 4.26 copies/μL input [8] [28] |
| Dynamic Range Precision (CV) | 7-11% [8] [28] | 6-13% [8] [28] |
| Impact of Restriction Enzymes | Minimal precision effect [8] [28] | Significant improvement with HaeIII vs. EcoRI [8] [28] |
| Accuracy (R² vs. expected concentration) | R²adj = 0.98 [8] [28] | R²adj = 0.99 [8] [28] |
The study demonstrated that while ddPCR showed a slightly lower LOD, ndPCR provided a better LOQ, indicating more reliable quantification at low concentrations [8] [28]. Both platforms showed high precision across dilution series, with ndPCR maintaining consistent coefficient of variation (CV) values (7-11%) compared to ddPCR's wider range (6-13%) [8] [28].
Multiplexing Capabilities and Applications
Multiplexing efficiency represents a critical differentiator between dPCR platforms for complex assays. ndPCR systems offer advanced multiplexing capabilities through two primary approaches:
- Spectral Multiplexing: Utilizes multiple fluorophores with distinct emission spectra. The QIAcuity platform, for example, offers six standard channels plus two hybrid channels for Long Stokes Shift (LSS) dyes, enabling detection of up to 8 targets [53].
- Amplitude Multiplexing: Simultaneously quantifies multiple targets in the same color channel by analyzing fluorescence intensity differences, potentially expanding multiplexing to 12 targets in a single reaction [53].
The applications for multiplex dPCR are extensive across research and clinical domains:
- Copy Number Variation (CNV) Analysis: Precisely determines target-to-reference gene ratios using duplex approaches [53].
- Rare Mutation Detection: Enables simultaneous quantification of somatic mutations and wild-type sequences [6] [53].
- Microbial Detection: Identifies multiple pathogens or resistance genes in a single reaction [53] [3].
- Cell and Gene Therapy: Supports vector copy number quantification and residual DNA detection [16] [53].
Throughput and Workflow Efficiency
Throughput considerations extend beyond mere sample numbers to include workflow integration, hands-on time, and total time to results:
Table 3: Throughput and Workflow Comparison
| Characteristic | Nanoplate dPCR | Droplet dPCR |
|---|---|---|
| Workflow Integration | Fully integrated system (partitioning, thermocycling, imaging) [30] [29] | Multiple instruments required [6] |
| Hands-on Time | Minimal; similar to qPCR workflow [6] [30] | Extensive; multiple transfer steps [6] |
| Total Time to Results | ~2 hours [30] [29] | 6-8 hours [6] [16] |
| Contamination Risk | Lower (closed system, minimal transfers) [6] [16] | Higher (multiple open processing steps) [6] |
| Automation Compatibility | High (standard plate formats) [30] [29] | Limited (specialized formats) [6] |
For quality control environments in cell and gene therapy manufacturing, the integrated workflow of ndPCR systems provides distinct advantages with their "sample-in, results-out" process that reduces hands-on time and contamination risks [16].
Experimental Protocols for Performance Validation
Protocol 1: Comparative Sensitivity and Precision Analysis
This protocol is adapted from the 2025 study comparing QIAcuity ndPCR and QX200 ddPCR platforms [8] [28].
Materials and Reagents:
- Synthetic oligonucleotides with known concentrations
- DNA from Paramecium tetraurelia cultures with defined cell counts
- Restriction enzymes (EcoRI and HaeIII)
- dPCR master mixes compatible with each platform
- Primers and probes for target genes
- QIAcuity Nanoplates (96-well) or ddPCR cartridges
Methodology:
- Sample Preparation: Create a dilution series of synthetic oligonucleotides covering 5-6 orders of magnitude. Prepare DNA extracts from Paramecium tetraurelia cell counts ranging from 10-1000 cells.
- Restriction Digestion: Treat half of the samples with EcoRI and the other half with HaeIII to evaluate enzyme effects on precision.
- Reaction Setup:
- For ndPCR: Load 40μL reactions into QIAcuity 96-well nanoplates
- For ddPCR: Prepare 20μL reactions for droplet generation
- Instrument Operation:
- ndPCR: Run on QIAcuity with integrated partitioning, thermocycling, and imaging
- ddPCR: Generate droplets, perform endpoint PCR, then read on droplet reader
- Data Analysis: Calculate LOD and LOQ using Poisson statistics. Determine precision via coefficient of variation (CV) across replicates.
Protocol 2: Multiplexing Efficiency Assessment
Materials and Reagents:
- DNA sample containing multiple targets of interest
- Multiplex probe PCR kit (e.g., QIAcuity High Multiplex Probe PCR Kit)
- Primers and probes for 5-12 targets with different fluorophores
- dPCR plates or cartridges compatible with chosen platform
Methodology:
- Assay Optimization: Initially optimize each assay individually for primer concentration and annealing temperature.
- Multiplex Reaction Setup: Combine all primers and probes in a single reaction mixture.
- Cross-talk Compensation: Establish a custom cross-talk matrix to address fluorescence bleed-between channels.
- Threshold Optimization: For amplitude multiplexing, set multiple thresholds within a single channel to distinguish between targets.
- Specificity Validation: Run no-template controls and single-positive controls to verify specific amplification.
- Data Analysis: Calculate copy numbers for each target and assess any amplification efficiency differences between singleplex and multiplex reactions.
The Scientist's Toolkit: Essential Research Reagents
Successful dPCR implementation requires careful selection of reagents and consumables. The following table outlines essential components for optimizing dPCR assays:
Table 4: Essential Digital PCR Research Reagents
| Reagent/Consumable | Function | Optimization Considerations |
|---|---|---|
| dPCR Master Mix | Provides enzymes, dNTPs, and buffer for amplification | Use platform-specific formulations; may require higher primer/probe concentrations than qPCR [54] |
| Hydrolysis Probes | Target-specific detection with fluorescent reporters | Double-quenched probes reduce background fluorescence; avoid excessive freeze-thaw cycles [54] |
| Restriction Enzymes | Fragment high-molecular-weight DNA for improved target accessibility | Enzyme selection affects precision; HaeIII may outperform EcoRI for some applications [8] [28] |
| dPCR Plates/Cartridges | Create partitions for single-molecule amplification | Choice affects partition numbers and volumes; nanoplates vs. droplet generators [6] |
| Positive Control Templates | Assay validation and optimization | Should match sample type (e.g., sheared DNA for cell-free DNA applications) [54] |
The choice between nanoplate and droplet digital PCR technologies depends heavily on specific application requirements and laboratory workflows. Nanoplate dPCR systems excel in environments prioritizing high-throughput processing, advanced multiplexing, and streamlined workflows with minimal hands-on time, making them particularly suitable for quality control settings in cell and gene therapy manufacturing [16]. Droplet dPCR systems offer exceptional partition numbers and established protocols with extensive literature support, potentially providing slight advantages in detection limits for some applications [8] [28].
For complex assays requiring simultaneous detection of multiple targets, ndPCR platforms currently offer superior multiplexing capabilities (up to 12-plex) compared to most ddPCR systems [53]. However, both technologies deliver excellent precision and accuracy when properly optimized, with recent comparative studies demonstrating comparable performance across most analytical parameters [8] [28].
Future developments in dPCR technology will likely focus on increasing multiplexing capacity, improving workflow automation, and reducing costs per sample. As these technologies evolve, ongoing comparative studies will remain essential for guiding researchers toward optimal platform selection based on their specific precision, throughput, and multiplexing requirements.
Data-Driven Decisions: Validating Performance Through Cross-Platform Comparative Studies
Digital PCR (dPCR) represents a significant advancement in nucleic acid quantification by enabling absolute target counting without the need for standard curves. This technology partitions a sample into thousands of individual reactions, statistically determining the target concentration based on the proportion of positive partitions using Poisson statistics [2]. As two principal partitioning methods have emerged—nanoplate-based (exemplified by the QIAGEN QIAcuity) and droplet-based (exemplified by the Bio-Rad QX200)—researchers require comprehensive, head-to-head comparisons of their performance characteristics. This guide objectively evaluates the precision, linear range, and practical application of these platforms based on recent comparative studies, providing scientists in research and drug development with critical data for platform selection.
Platform Performance at a Glance
The following table synthesizes key performance metrics from direct comparative studies, offering a quantitative overview of the two platforms.
Table 1: Direct Performance Comparison of Nanoplate-based and Droplet-based dPCR Platforms
| Performance Parameter | Nanoplate-based dPCR (QIAcuity) | Droplet-based dPCR (QX200) |
|---|---|---|
| Lower Limit of Detection (LOD) | 0.39 copies/µL [55] | 0.17 copies/µL [55] |
| Lower Limit of Quantification (LOQ) | 1.35 copies/µL [55] | 4.26 copies/µL [55] |
| Typical Partition Count | ~26,000 partitions [19] [20] | ~20,000 droplets [16] [56] |
| Precision (from Oligo Dilutions) | CV: 7-11% [55] | CV: 6-13% [55] |
| Precision (from Cell DNA, EcoRI) | CV range: 0.6% - 27.7% [55] | CV range: 2.5% - 62.1% [55] |
| Precision (from Cell DNA, HaeIII) | CV range: 1.6% - 14.6% [55] | CV improved to <5% for all cell numbers [55] |
| Dynamic Range (from HAdV study) | Quantification from 0.95 to 770 copies/µL [7] | Information not available in search results |
| Workflow Time | < 90 minutes [16] | 6-8 hours [16] |
Experimental Insights: Key Comparative Studies
Copy Number Analysis in Protists
A 2025 study provided a direct comparison using synthetic oligonucleotides and DNA from the ciliate Paramecium tetraurelia to evaluate the platforms' precision and accuracy in quantifying gene copies [55].
- Experimental Protocol: Researchers tested both platforms using a dilution series of synthetic oligonucleotides and DNA extracted from a known number of ciliate cells (1 to 100 cells). The study design included testing two different restriction enzymes (EcoRI and HaeIII) to assess their impact on the accessibility of tandemly repeated gene targets. For each sample and condition, the measured gene copy number was compared against the expected value to determine accuracy, while precision was evaluated by calculating the Coefficient of Variation (CV) across replicates [55].
- Key Findings on Precision: Both platforms demonstrated high precision (CVs < 13%) across most synthetic oligonucleotide dilutions. However, when analyzing complex genomic DNA from ciliates, the choice of restriction enzyme significantly impacted precision, especially for the droplet-based system (QX200). Using HaeIII instead of EcoRI dramatically improved the precision of the QX200, bringing all CVs below 5%. The nanoplate system (QIAcuity) showed less variability due to enzyme choice [55].
- Key Findings on Linearity and Accuracy: Both platforms exhibited a strong linear relationship between expected and measured gene copies from synthetic targets (R²adj > 0.98). However, both systems consistently reported measured gene copies lower than expected, with this underestimation being slightly more pronounced in the nanoplate system at increasing concentrations [55].
GMO Quantification in Food and Feed
Another 2025 study compared the two platforms for the duplex quantification of genetically modified organisms (GMOs) in soybean, a critical application for food safety and labeling compliance [20].
- Experimental Protocol: The study performed an in-house validation of duplex dPCR methods for detecting two GM soybean events (MON-04032-6 and MON89788) with the lectin reference gene. Identical primer-probe sets were used on both platforms. DNA was extracted from certified reference materials, and performance was assessed through parameters including specificity, dynamic range, limit of quantification, and accuracy (trueness and precision) [20].
- Key Findings: The validation data for both the nanoplate and droplet platforms agreed with acceptance criteria according to international guidance documents. The duplex methods on both platforms demonstrated performance equivalent to the established singleplex real-time PCR method, confirming their suitability for a full collaborative trial validation [20].
Workflow and Practical Implementation
The fundamental difference between the two platforms lies in their partitioning mechanisms, which directly impacts laboratory workflow and practicality.
The nanoplate system utilizes a microfluidic chip with fixed wells, integrating partitioning, thermocycling, and imaging into a single, automated instrument. In contrast, the droplet system requires generating a water-in-oil emulsion, transferring droplets to a PCR plate, and then reading them in a separate instrument [16] [2]. This streamlined workflow of the nanoplate system reduces hands-on time from several hours to under 90 minutes and minimizes contamination risk and human error, making it particularly advantageous for quality control (QC) environments [16].
Essential Reagents for Robust dPCR
The following table lists key reagents and their functions, as utilized in the cited comparative studies.
Table 2: Research Reagent Solutions for dPCR Experiments
| Reagent / Material | Function | Application Example |
|---|---|---|
| Restriction Enzymes (e.g., HaeIII) | Digest genomic DNA to improve target accessibility and precision [55]. | Greatly improved precision in ciliate gene copy number analysis, especially for ddPCR [55]. |
| dPCR Supermix | Optimized buffer, enzymes, and dNTPs for efficient amplification in partitions [56]. | Used in the validation of vector copy number in CAR/TCR T-cell products [56]. |
| Fluorophore-Led Probes (e.g., FAM, HEX) | Target-specific detection with different fluorescent dyes for multiplexing [19] [7]. | Enabled duplex detection of pork and chicken in meat products [19] and HAdV detection [7]. |
| Certified Reference Materials (CRMs) | Provide known, standardized target quantities for method validation [20]. | Essential for the in-house validation of GMO quantification methods [20]. |
Direct comparative studies indicate that both nanoplate and droplet dPCR platforms deliver excellent precision and a wide linear range for nucleic acid quantification, often outperforming quantitative real-time PCR [55] [26]. The choice between them should be guided by specific application needs. The droplet system (QX200) may offer a marginally superior lower Limit of Detection [55], while the nanoplate system (QIAcuity) demonstrates a robust and streamlined workflow beneficial for high-throughput and regulated environments [16]. Factors such as sample type and preparation, including the strategic use of restriction enzymes, are critical for achieving optimal precision with complex samples on either platform [55]. As the technology evolves, both platforms are poised to become indispensable tools for precise molecular quantification in research, clinical diagnostics, and quality control.
This guide provides an objective comparison of the performance of nanoplate-based digital PCR (dPCR) and droplet digital PCR (ddPCR) platforms, framed within a broader thesis on measurement precision and accuracy in genetic research.
Digital PCR (dPCR) represents a significant advancement in nucleic acid quantification by enabling absolute measurement of target sequences without the need for a standard curve. [15] [57] The core principle involves partitioning a PCR reaction into thousands of individual reactions, so that each contains zero, one, or a few target molecules. After end-point PCR amplification, the fraction of positive partitions is counted, and the absolute concentration of the target nucleic acid is calculated using Poisson statistics. [58] [15] [57] This approach provides exceptional precision and sensitivity, particularly for applications requiring detection of small copy number variations or rare genetic events. [59]
Two major partitioning methodologies have emerged: droplet-based dPCR (ddPCR), which generates thousands of nanoliter-sized water-in-oil droplets, and nanoplate-based dPCR, which utilizes microfluidics to distribute samples into fixed nanowells on a chip. [15] While both aim to achieve precise single-molecule detection, their underlying technologies create meaningful differences in performance characteristics, workflow efficiency, and application suitability that researchers must consider for specific genetic analysis scenarios.
Performance Comparison: Nanoplate vs. Droplet dPCR
Quantitative Performance Metrics
Direct comparative studies provide valuable insights into the operational differences between these platforms. A 2025 study systematically compared the QIAcuity One nanoplate dPCR system (QIAGEN) with the QX200 droplet dPCR system (Bio-Rad) for copy number analysis in the protist Paramecium tetraurelia. [8]
Table 1: Performance Metrics from Protist Copy Number Analysis [8]
| Performance Parameter | Nanoplate dPCR (QIAcuity One) | Droplet dPCR (QX200) |
|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL input | 0.17 copies/µL input |
| Limit of Quantification (LOQ) | 1.35 copies/µL input | 4.26 copies/µL input |
| Precision (with EcoRI enzyme) | CV: 0.6% - 27.7% | CV: 2.5% - 62.1% |
| Precision (with HaeIII enzyme) | CV: 1.6% - 14.6% | CV: < 5% (all samples) |
| Reaction Volume | 40 µL | 20 µL |
| Absolute Copies per Reaction (at LOQ) | 54 copies | 85.2 copies |
The data reveals a trade-off between sensitivity and precision. While the droplet system demonstrated a slightly better (lower) Limit of Detection, the nanoplate system showed a superior (lower) Limit of Quantification, indicating potentially better performance at low concentration levels requiring precise measurement. [8] Notably, restriction enzyme choice significantly impacted precision, especially for the droplet system, where switching from EcoRI to HaeIII dramatically improved precision (CV from >60% to <5%). This highlights how assay optimization remains crucial for both platforms. [8]
Platform Workflow and Usability
Beyond pure quantification metrics, practical workflow differences significantly impact laboratory efficiency.
Table 2: Workflow and Practical Comparison [8] [19]
| Feature | Nanoplate dPCR | Droplet dPCR |
|---|---|---|
| Partitioning Method | Microfluidics into fixed nanowells | Water-oil emulsion droplet generation |
| Typical Partitions | ~26,000 nanowells [3] [19] | ~20,000 droplets [60] |
| Workflow | Integrated partitioning, thermocycling, and imaging [19] | Multiple steps: droplet generation, transfer, thermocycling, droplet reading [19] |
| Automation | High, single-step procedure [19] | Lower, requires manual or instrument transfer |
| Risk of Contamination/Droplet Shearing | Lower (closed system) [19] | Higher [19] |
| Technical Skill Required | Lower, similar to qPCR [19] | Higher, requires specialized pipetting skills [19] |
The nanoplate system offers a more streamlined and automated workflow, reducing hands-on time and the potential for user error. [19] Its "all-in-one" nature minimizes the risk of partition loss or cross-contamination, which can occur during the multiple transfer steps of traditional droplet systems. [19]
Experimental Protocols for Platform Comparison
The following detailed methodology is adapted from the 2025 comparative study of dPCR platforms in protist copy number analysis, which serves as a model for robust cross-platform evaluation. [8]
Sample Preparation and DNA Extraction
- Biological Material: Use a well-characterized model organism. The referenced study used varying cell numbers of the ciliate Paramecium tetraurelia. [8]
- DNA Extraction: Perform standard phenol-chloroform or kit-based nucleic acid extraction from the biological samples.
- DNA Quantification: Quantify the extracted DNA using a fluorometer. Note that concentrations may deviate from manufacturer specifications for synthetic controls, as was observed in the reference study. [8]
- Restriction Digestion: To improve accessibility of tandemly repeated genes, digest DNA with restriction enzymes. Test multiple enzymes (e.g., EcoRI and HaeIII) as their efficiency can significantly impact precision and copy number estimates. [8]
dPCR Assay Setup and Execution
For Nanoplate dPCR (QIAcuity One):
- Reaction Mix: Prepare a PCR master mix containing the dPCR reaction buffer, primers, probes (e.g., EvaGreen or TaqMan), and the template DNA.
- Loading: Pipette the reaction mix into the wells of a dedicated nanoplate.
- Run: Place the nanoplate into the QIAcuity One instrument. The instrument automatically performs partitioning into ~26,000 nanowells, PCR thermocycling, and fluorescence imaging. [3] [19]
For Droplet dPCR (QX200):
- Reaction Mix: Prepare a PCR master mix as above.
- Droplet Generation: Transfer the reaction mix into the DG8 cartridge. Using the QX200 Droplet Generator, the aqueous sample is emulsified into approximately 20,000 nanoliter-sized oil droplets. [60]
- Transfer and Sealing: Carefully transfer the generated droplets to a 96-well PCR plate and seal it.
- Thermocycling: Perform end-point PCR amplification on a conventional thermal cycler.
- Droplet Reading: Place the PCR plate into the QX200 Droplet Reader, which flows droplets one-by-one through a fluorescence detector to identify positive and negative droplets. [8]
Data Analysis
- Threshold Setting: For both platforms, use the manufacturer's software to set fluorescence amplitude thresholds that distinguish positive partitions from negative partitions.
- Concentration Calculation: The software automatically applies Poisson statistics to the fraction of positive partitions to calculate the absolute concentration of the target in copies per microliter (copies/μL). [58] [15]
- Statistical Comparison: Compare the calculated concentrations from both platforms against expected values (for synthetic controls) or against each other (for biological samples). Key metrics include accuracy (deviation from expected), precision (Coefficient of Variation, %CV), Limit of Detection (LOD), and Limit of Quantification (LOQ). [8]
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for dPCR Comparison Studies
| Item | Function | Example from Case Study |
|---|---|---|
| dPCR Platform | Instrument for partitioning, amplification, and analysis. | QIAcuity One (QIAGEN); QX200 (Bio-Rad) [8] |
| Assay Chemistry | Fluorescent probes or dyes for target-specific detection. | EvaGreen dye or TaqMan probes [8] |
| Restriction Enzymes | Digest DNA to improve access to target genes, especially in repeats. | HaeIII, EcoRI [8] |
| Nanoplates/Cartridges | Consumables for sample partitioning. | QIAcuity nanoplate (QIAGEN); DG8 Cartridge (Bio-Rad) [8] [19] |
| Template DNA | The nucleic acid sample of interest for quantification. | Genomic DNA from Paramecium tetraurelia; synthetic oligonucleotides [8] |
Application in Broader Genetic Research
The precision of dPCR makes it invaluable beyond protist research, with critical applications in human genetics and drug development.
- Human Oncology and Liquid Biopsy: dPCR excels at detecting rare mutations and quantifying copy number variations (CNVs) in complex backgrounds. It is 100 times more sensitive than conventional methods for rare mutation detection and can quantify circulating tumor DNA (ctDNA) in liquid biopsies for monitoring therapeutic response and tumor burden. [59]
- Infectious Disease Diagnostics: dPCR demonstrates superior accuracy for quantifying viral loads, particularly for medium to high concentrations of pathogens like influenza, RSV, and SARS-CoV-2. Its tolerance to inhibitors found in clinical samples (e.g., mucus) makes it robust for direct pathogen detection in respiratory samples. [3]
- Cell and Gene Therapy Development: dPCR is used for the absolute quantification of viral vector titers (e.g., AAV), monitoring vector biodistribution, and detecting genome editing events (e.g., CRISPR-Cas9), providing essential quality control and efficacy data. [59] [61]
- Food Authentication and Environmental Surveillance: The high sensitivity of nanoplate dPCR enables the detection of food adulteration, such as identifying pork and chicken in processed meats with a limit of detection of 0.1%. It also accurately measures pathogens in environmental samples like wastewater. [59] [19]
This comparison reveals that the choice between nanoplate and droplet dPCR is application-dependent. Nanoplate dPCR (QIAcuity One) offers a more automated, user-friendly workflow with higher precision in quantified low-concentration samples and greater resilience to practical handling errors. [8] [19] Droplet dPCR (QX200) may provide a marginally better limit of detection. [8]
Both technologies, however, deliver the core advantage of dPCR: absolute quantification of nucleic acids with high precision and accuracy, enabling advancements across diverse fields from fundamental protist genetics to clinical human diagnostics and therapeutic development.
The accurate detection of DNA methylation in clinical samples is crucial for identifying novel epigenetic biomarkers in breast cancer. Formalin-fixed, paraffin-embedded (FFPE) tissues present particular challenges for molecular analysis due to DNA fragmentation and degradation. This case study objectively compares the performance of two digital PCR platforms—nanoplate-based dPCR and droplet-based ddPCR—in detecting CDH13 gene methylation in FFPE breast cancer tissues, providing experimental data to inform platform selection for molecular diagnostics [5] [62].
Experimental Design and Methodology
Sample Collection and Preparation
The study analyzed 141 FFPE breast cancer tissue samples provided by the Department of Pathological Anatomy, Jessenius Faculty of Medicine in Martin [5]. Genomic DNA was isolated from deparaffinized tissue using a DNeasy Blood and Tissue kit (Qiagen), with DNA concentration measured using Qubit 3.0 with a dsDNA BR Assay kit (Thermo Fisher Scientific) [5]. For methylation analysis, 1 µg of isolated DNA was modified with an EpiTect Bisulfite kit (Qiagen) following manufacturer instructions [5].
Target Selection and Assay Design
The study focused on methylation status of three CpG sites in the CDH13 promoter region (chr16:82,626,843; chr16:82,626,845; chr16:82,626,859) [5]. CDH13 was selected as it was identified as the most frequently methylated gene in a cohort of Slovak patients diagnosed with invasive ductal carcinoma [62]. Primers and probes were designed using MethPrimer and Primer3Plus online programs [5]. The assay was optimized for simultaneous detection of methylated and unmethylated DNA in a single reaction mix using FAM-labeled methylated-specific and HEX-labeled unmethylated-specific probes [5].
Table 1: Primer and Probe Sequences for CDH13 Methylation Analysis
| Component | Sequence (5' → 3') |
|---|---|
| Forward primer | AAAGAAGTAAATGGGATGTTATTTTC |
| Reverse primer | ACCAAAACCAATAACTTTACAAAAC |
| M-Probe (FAM) | TCGCGAGGTGTTTATTTCGT |
| UnM-Probe (HEX) | TTTTGTGAGGTGTTTATTTTGTATTTGT |
Platform-Specific Protocols
QIAcuity Digital PCR (Nanoplate-based)
Each dPCR reaction was prepared in a 12 μL volume containing 3 µL of QIAcuity 4× Probe PCR master mix, 0.96 µL of forward/reverse primer, 0.48 µL of each probe, 2.5 µL of DNA template, and RNase-free water [5]. Reaction mixtures were pipetted into 24-well nanoplates (8,500 partitions per well) and processed in QIAcuity One [5]. Cycling conditions consisted of initial heat activation (95°C for 2 min), followed by 40 cycles of denaturation (95°C for 15 s) and a combined annealing/extension step (57°C for 1 min) [5].
QX200 Droplet Digital PCR (Droplet-based)
The ddPCR reaction mixture contained 10 μL of Supermix for Probes (No dUTP) (Bio-Rad), 0.45 μL of forward/reverse primer, 0.45 μL of each probe, 2.5 μL of DNA template, and RNase-free water to a final volume of 20 μL [5]. Approximately 20,000 droplets per sample were generated using the QX200 Droplet Generator [5]. The optimized PCR protocol consisted of an initial denaturation (95°C for 10 min), 40 cycles of denaturation (94°C for 30 s), and combined annealing/extension (57°C for 1 min) [5].
Data Analysis
For both platforms, methylation level was expressed as a ratio of positive FAM-detected partitions (methylated) to the sum of all positive partitions detected on both channels (methylated + unmethylated) [5]. Acceptance criteria required over 7,000 valid partitions and at least 100 positive partitions for nanoplate-based dPCR [5].
Experimental Workflow for Methylation Analysis. The diagram illustrates the standardized sample processing and platform-specific partitioning for CDH13 methylation detection in FFPE breast cancer tissues.
Comparative Performance Analysis
Analytical Performance Metrics
Both platforms demonstrated excellent performance for CDH13 methylation detection in FFPE samples, with strong correlation between measurements [5].
Table 2: Analytical Performance of dPCR Platforms for CDH13 Methylation Detection
| Performance Metric | QIAcuity dPCR (Nanoplate) | QX200 ddPCR (Droplet) |
|---|---|---|
| Specificity | 99.62% | 100% |
| Sensitivity | 99.08% | 98.03% |
| Correlation between platforms | r = 0.954 | r = 0.954 |
| Partitions per reaction | 8,500 | ~20,000 |
| Reaction volume | 12 μL | 20 μL |
| Methylation quantification | Ratio of FAM-positive to total positive partitions | Ratio of FAM-positive to total positive partitions |
Methodological Advantages for Methylation Analysis
Both dPCR platforms offer significant advantages over conventional methylation analysis methods. Compared to semiquantitative methods like MS-MLPA, dPCR provides higher precision and technical simplicity for measuring methylation of target CpGs [62]. Digital PCR enables absolute quantification without external references and demonstrates greater robustness to PCR efficiency variations compared to real-time PCR [5]. This enhanced precision is particularly valuable for detecting rare methylation events in degraded DNA from FFPE material [5] [62].
Technical Considerations for Platform Selection
Workflow and Practical Implementation
Substantial differences exist in the practical implementation of nanoplate versus droplet dPCR systems, impacting their suitability for different laboratory environments.
Table 3: Workflow Comparison Between dPCR Platforms
| Parameter | QIAcuity dPCR (Nanoplate) | QX200 ddPCR (Droplet) |
|---|---|---|
| Partitioning mechanism | Fixed nanoplate array | Water-oil emulsion droplets |
| Hands-on time | Minimal | Multiple manual steps |
| Automation level | Integrated "sample-to-result" system | Multiple instruments required |
| Time to results | < 90 minutes | 6-8 hours |
| Risk of contamination | Lower (closed system) | Higher (multiple transfers) |
| Multiplexing capability | Available for 4-12 targets | Limited in standard systems |
The nanoplate-based system offers a streamlined "sample-in, results-out" process on a single instrument, significantly reducing hands-on time and potential for human error [16]. This integrated approach inherently lowers contamination risk, a critical factor for maintaining sample integrity in quality control environments [16]. In contrast, droplet-based systems require multiple instruments and manual transfers, extending the total workflow time to 6-8 hours [16].
Application-Specific Performance Considerations
While both platforms demonstrated excellent analytical performance for CDH13 methylation detection, selection criteria may depend on specific research requirements. A comparative study of different digital PCR platforms highlighted that both can achieve high precision, though performance can be influenced by factors such as restriction enzyme choice in assay design [28]. The number of partitions differs substantially between platforms (8,500 for nanoplate vs. ~20,000 for droplet), potentially affecting detection sensitivity for very rare methylation events [5].
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagent Solutions for dPCR Methylation Analysis
| Reagent / Kit | Manufacturer | Function in Workflow |
|---|---|---|
| DNeasy Blood & Tissue Kit | Qiagen | DNA isolation from FFPE tissue |
| EpiTect Bisulfite Kit | Qiagen | Conversion of unmethylated cytosines to uracils |
| QIAcuity 4× Probe PCR Master Mix | Qiagen | Reaction mix for nanoplate-based dPCR |
| Supermix for Probes (No dUTP) | Bio-Rad Laboratories | Reaction mix for droplet-based dPCR |
| Droplet Generation Oil for Probes | Bio-Rad Laboratories | Oil for water-in-oil emulsion droplet formation |
| Fully methylated/unmethylated DNA controls | Qiagen | Assay validation and control samples |
This case study demonstrates that both nanoplate-based dPCR and droplet-based ddPCR platforms provide highly sensitive and specific detection of CDH13 methylation in FFPE breast cancer tissues, with strong correlation between measurements [5]. The choice between platforms should be guided by specific research needs: nanoplate systems offer advantages in workflow efficiency, automation, and reduced contamination risk, while droplet systems provide higher partition numbers [5] [16]. Both technologies represent significant advancements over conventional methylation analysis methods, offering absolute quantification with high precision and robustness for biomarker analysis in clinically relevant samples [5] [62].
The accurate quantification of Genetically Modified Organisms (GMOs) in food and feed samples represents a critical challenge in molecular analytics, with implications for regulatory compliance, labeling accuracy, and consumer safety. Digital PCR (dPCR) has emerged as a powerful third-generation PCR technique that enables absolute quantification of nucleic acids without requiring standard curves, offering significant advantages for GMO testing [16]. Among dPCR technologies, two primary platforms have gained prominence: droplet digital PCR (ddPCR) which employs water-oil emulsion to create thousands of nanoliter-sized droplets, and nanoplate-based digital PCR (ndPCR) which distributes samples across fixed microwells [16] [19]. This case study provides an objective comparison of these platforms within the broader thesis on precision and accuracy in dPCR research, examining their performance characteristics specifically relevant to GMO quantification applications.
Performance Comparison: Nanoplate vs. Droplet dPCR
Key Performance Metrics
Evaluation of platform performance for molecular quantification requires assessment of multiple technical parameters as summarized in Table 1.
Table 1: Performance comparison of nanoplate and droplet digital PCR platforms
| Parameter | Nanoplate dPCR | Droplet dPCR | Experimental Context |
|---|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL [28] [8] | 0.17 copies/µL [28] [8] | Synthetic oligonucleotides |
| Limit of Quantification (LOQ) | 1.35 copies/µL [28] [8] | 4.26 copies/µL [28] [8] | Synthetic oligonucleotides |
| Precision (CV Range) | 0.6%-27.7% (with EcoRI); 1.6%-14.6% (with HaeIII) [28] [8] | 2.5%-62.1% (with EcoRI); <5% (with HaeIII) [28] [8] | Paramecium tetraurelia DNA |
| Dynamic Range | 0.9476 to 770.4 copies/μL [43] | <0.5 to >3000 copies/μL [28] [8] | Human adenovirus DNA; synthetic oligonucleotides |
| Sensitivity in Food Matrices | 0.1% (w/w) for meat species [19]; 0.1 mg/kg for sesame [22] | 1% (w/w) for meat species [19] | Processed meat products; sesame in dough/biscuits |
| Partition Number | ~24,000-26,000 [19] | ~20,000 [16] | Standard instrument specifications |
| Multiplexing Capability | Available in 4-12 targets [16] | Limited, though newer models detect up to 12 targets [16] | Platform specifications |
Platform Workflow and Operation
The fundamental difference between nanoplate and droplet dPCR platforms lies in their partitioning mechanisms and associated workflows, which significantly impact their practical implementation in analytical laboratories.
Table 2: Workflow and operational comparison
| Aspect | Nanoplate dPCR | Droplet dPCR |
|---|---|---|
| Partitioning Mechanism | Fixed array/nanoplate [16] | Emulsion droplets [16] |
| Time to Results | <90 minutes [16] | 6-8 hours [16] |
| Workflow Steps | Single integrated instrument [19] | Multiple instruments and transfer steps [19] |
| Contamination Risk | Minimal (closed system) [43] | Higher (multiple open steps) [19] |
| Ease of Use | Integrated automated system [16] | Requires specialized pipetting skills [19] |
| Ideal Setting | QC environment [16] | Development labs [16] |
Figure 1: Comparative workflows for nanoplate and droplet digital PCR platforms highlighting the streamlined process of nanoplate systems versus multiple manual steps in droplet-based approaches.
Experimental Protocols for GMO Quantification
Sample Preparation and DNA Extraction
For GMO quantification in food and feed samples, proper sample preparation is paramount. DNA should be extracted from representative samples of approximately 100-200 mg of homogenized material using validated extraction kits suitable for complex matrices [19]. The DNA concentration should be quantified using fluorometric methods (e.g., Qubit Fluorometer) to ensure accurate measurement, as spectrophotometric methods may be influenced by contaminants [43]. DNA purity should be assessed through A260/A280 and A260/A230 ratios, with acceptable ranges between 1.8-2.0 and 2.0-2.2, respectively. For samples with suspected PCR inhibitors, additional purification steps using inhibitor removal kits may be necessary [27].
Digital PCR Assay Design
GMO quantification requires careful assay design targeting specific transgenic elements. The protocol should include:
Primer and Probe Design: Design primers and hydrolysis probes (e.g., TaqMan) targeting event-specific transgenic sequences (e.g., 35S promoter, NOS terminator) alongside an endogenous reference gene for normalization [19]. In silico validation should be performed using BLAST tools to ensure specificity [43].
Reaction Setup: Prepare 20-40µL reactions containing 1× dPCR master mix, 900 nM of each primer, 250 nM of probe, and DNA template [27] [43]. Optimal DNA input should be determined empirically but typically ranges from 1-100 ng total DNA depending on the expected GMO percentage.
Restriction Enzyme Digestion: For targets with potential tandem repeats or complex structures, incorporate restriction enzymes (e.g., HaeIII) to improve DNA accessibility and precision. Studies have demonstrated that enzyme selection significantly impacts precision, particularly for ddPCR [28] [8].
Platform-Specific Protocols
Nanoplated dPCR Protocol
For nanoplate-based systems (e.g., QIAcuity):
Partitioning: Load the reaction mixture into designated nanoplates and perform partitioning using the integrated instrument [19]. The QIAcuity system typically generates >24,000 partitions [19].
Amplification: Perform PCR cycling with conditions optimized for the assay: initial denaturation at 95°C for 2 minutes, followed by 40-45 cycles of denaturation at 95°C for 15 seconds and annealing/extension at 60°C for 60 seconds [43].
Imaging and Analysis: The integrated imaging system automatically captures fluorescence data from each partition, and software calculates the target concentration (copies/μL) using Poisson statistics [19].
Droplet dPCR Protocol
For droplet-based systems (e.g., Bio-Rad QX200):
Droplet Generation: Transfer the reaction mixture to the droplet generator cartridge alongside droplet generation oil to create approximately 20,000 nanoliter-sized droplets [16].
Transfer and Amplification: Carefully transfer the emulsion to a 96-well PCR plate, seal, and perform endpoint PCR with cycling conditions similar to ndPCR [27].
Droplet Reading and Analysis: Transfer the plate to a droplet reader which counts positive and negative droplets, with subsequent calculation of target concentration using Poisson statistics [27].
Data Analysis and Validation
For GMO quantification, calculate the percentage using the formula: %GMO = (Transgene concentration / Endogenous reference gene concentration) × 100
Method validation should include determination of limit of detection (LOD), limit of quantification (LOQ), precision (coefficient of variation), and dynamic range using certified reference materials when available [43].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential reagents and materials for dPCR-based GMO quantification
| Item | Function | Examples/Specifications |
|---|---|---|
| DNA Extraction Kit | High-quality DNA isolation from complex matrices | Kits optimized for processed foods; inhibitor removal technology |
| dPCR Master Mix | Amplification reaction foundation | One-step RT-ddPCR Advanced Kit; probe-based chemistries |
| Sequence-Specific Primers | Target amplification | Designed to detect specific transgenic elements (e.g., 35S promoter) |
| Hydrolysis Probes | Target-specific detection | FAM-labeled probes with appropriate quenchers |
| Restriction Enzymes | Enhance DNA accessibility | HaeIII, EcoRI - improves precision particularly for complex targets |
| Digital PCR System | Platform for partitioning and analysis | QIAcuity (nanoplate); QX200/QX600 (droplet) |
| Reference Materials | Method validation | Certified reference materials with known GMO percentages |
| Microcentrifuge Tubes | Sample preparation | Low DNA binding, nuclease-free |
| Filter Tips | Contamination prevention | Aerosol-resistant for critical pipetting steps |
Discussion
Application to GMO Quantification
The performance characteristics of both dPCR platforms present distinct advantages for GMO quantification. The superior sensitivity of ndPCR, demonstrated by its ability to detect 0.1% (w/w) adulteration in meat species [19] and 0.1 mg/kg sesame in food matrices [22], translates directly to GMO detection where regulatory thresholds often mandate quantification at 0.1-0.9% levels. This enhanced sensitivity, coupled with reduced contamination risk through closed systems [43], makes ndPCR particularly valuable for routine testing laboratories requiring robust, high-throughput methods for regulatory compliance.
The observed improvement in precision with specific restriction enzymes (e.g., HaeIII) for both platforms [28] [8] highlights a critical methodological consideration for GMO quantification, particularly when analyzing processed foods where DNA integrity may be compromised. This effect was more pronounced for ddPCR, where restriction enzyme selection dramatically improved precision from >60% CV to <5% CV [28] [8], suggesting that methodological optimization may mitigate some platform limitations.
Platform Selection Criteria
Selection between nanoplate and droplet dPCR platforms for GMO quantification should be guided by application-specific requirements:
Nanoplate dPCR is optimal for: Routine quality control environments requiring rapid results (<90 minutes) [16], high-throughput testing with minimal hands-on time, applications demanding enhanced sensitivity for low-level detection [19] [22], and regulated environments where contamination control and workflow standardization are prioritized [16].
Droplet dPCR remains valuable for: Research and development settings requiring maximal flexibility, applications where lower LOD is critical [28] [8], and laboratories with established expertise in droplet-based methodologies.
This comparative analysis demonstrates that both nanoplate and droplet digital PCR platforms offer robust solutions for GMO quantification in food and feed samples, with the optimal choice dependent on specific application requirements. Nanoplate systems provide distinct advantages in workflow efficiency, sensitivity for trace detection, and practical implementation in quality control environments, while droplet systems offer marginally superior detection limits. Both platforms deliver the precision and accuracy required for compliance with regulatory thresholds, with performance further enhanced through methodological optimization including restriction enzyme selection. As dPCR technologies continue to evolve, their implementation in GMO testing laboratories will strengthen the reliability of molecular quantification for food and feed safety monitoring.
The Workflow and Cost-Benefit Analysis for Research vs. Regulated Environments
Digital PCR (dPCR) has emerged as a powerful tool for the absolute quantification of nucleic acids, offering superior precision and sensitivity over traditional quantitative PCR (qPCR) by partitioning samples into thousands of individual reactions [58]. Within the dPCR ecosystem, two primary partitioning technologies have come to the fore: droplet digital PCR (ddPCR) and nanoplate-based dPCR (ndPCR). The core difference lies in the method of partitioning; ddPCR uses a water-oil emulsion to generate thousands of nanoliter-sized droplets, while ndPCR distributes the sample across a fixed plate containing thousands of micro-wells or nanoplanes [16] [6]. This fundamental distinction drives significant differences in workflow, performance, and suitability for various environments. For researchers, scientists, and drug development professionals, the choice between these platforms is not merely technical but strategic, impacting efficiency, cost, and compliance. This guide provides an objective comparison of ddPCR and ndPCR, framing the analysis within the critical context of application-specific requirements for research versus regulated environments, supported by experimental data and a detailed cost-benefit analysis.
Technology Comparison: Partitioning Mechanisms and Workflows
Fundamental Operating Principles
- Droplet Digital PCR (ddPCR): This method relies on generating an emulsion of thousands to millions of nanoliter- or picoliter-sized water-in-oil droplets, which act as independent reaction chambers [6] [58]. The nucleic acid sample is randomly distributed across these droplets. After end-point PCR amplification, each droplet is analyzed in a flow cytometer-like droplet reader to count the positive and negative reactions, with absolute quantification calculated using Poisson statistics [58].
- Nanoplate-based Digital PCR (ndPCR): This approach utilizes a microfluidic chip or nanoplate containing a fixed array of nanowells—typically thousands to tens of thousands per sample [29] [6]. The PCR reaction mix is loaded onto the nanoplate, which is then sealed. The entire plate undergoes thermal cycling, and fluorescence is measured simultaneously for all partitions using an imaging system [63]. This "all-in-one" instrument design integrates partitioning, thermocycling, and imaging [29].
Workflow Comparison: A Step-by-Step Analysis
The workflow divergence between the two technologies is a critical differentiator. The following diagram summarizes the key steps for each platform.
Key Workflow Differences:
- Instrument Integration: The ndPCR workflow is notably more streamlined, with partitioning, thermocycling, and imaging occurring within a single, integrated instrument [29] [6]. This reduces hands-on time and the potential for human error.
- Manual Steps: The ddPCR workflow involves multiple instruments (droplet generator, thermal cycler, droplet reader) and requires manual transfer steps (e.g., transferring the emulsion to a PCR plate after generation) [64] [6]. This increases hands-on time and the risk of contamination or droplet shearing.
- Time Efficiency: The complete ndPCR workflow, from sample loading to results, can be completed in less than 2 hours [63]. In contrast, a typical ddPCR workflow can take between 6-8 hours due to its multiple, sequential steps [16].
- Ease of Use: The ndPCR workflow is often compared to the familiar qPCR process, making it easier to adopt with minimal training [6]. The ddPCR workflow requires more specialized training to master droplet handling and avoid pitfalls like coalescence or shearing [6].
Performance and Precision: Experimental Data Comparison
While both technologies are capable of highly precise quantification, independent studies have directly compared their performance in controlled settings.
Sensitivity and Limits of Detection
A 2025 study in Scientific Reports compared the QIAcuity One (ndPCR) and Bio-Rad QX200 (ddPCR) platforms using synthetic oligonucleotides and ciliate DNA. The results demonstrated that both platforms are suitable for sensitive applications, with slightly different limits of detection (LOD) and quantification (LOQ) [8].
Table 1: Comparison of LOD and LOQ between ndPCR and ddPCR [8]
| Platform | Limit of Detection (LOD) | Limit of Quantification (LOQ) | Notes |
|---|---|---|---|
| Nanoplate dPCR (QIAcuity One) | 0.39 copies/µL input | 1.35 copies/µL input | Based on a 40 µL reaction volume |
| Droplet ddPCR (QX200) | 0.17 copies/µL input | 4.26 copies/µL input | Based on a 20 µL reaction volume |
The study concluded that both platforms showed "similar detection and quantification limits" and "high precision across most analyses" [8]. The choice of restriction enzyme was also found to impact precision, particularly for the ddPCR system [8].
Performance in Viral Detection and Quantification
Digital PCR consistently demonstrates superior sensitivity and accuracy compared to qPCR, particularly for low viral loads. A 2024 study on rice black-streaked dwarf virus (RBSDV) found that reverse transcription dPCR (RT-dPCR) on a QIAcuity platform had a detection limit of 0.096 copies/µL, which was approximately 100 times more sensitive than RT-qPCR [65]. Similarly, a 2025 study on respiratory viruses (Influenza, RSV, SARS-CoV-2) found that dPCR "demonstrated superior accuracy, particularly for high viral loads," and showed "greater consistency and precision" compared to Real-Time RT-PCR [3].
The Impact of Partition Number and Volume
The following diagram illustrates the relationship between partition characteristics and the resulting data output for each platform, which underpins their performance.
The Scientist's Toolkit: Essential Reagents and Materials
A successful dPCR experiment, regardless of platform, relies on a core set of reagents and materials. The table below details these essential components.
Table 2: Essential Research Reagent Solutions for Digital PCR
| Item | Function | Platform Notes |
|---|---|---|
| dPCR Master Mix | Provides enzymes, dNTPs, and buffer optimized for the digital PCR reaction. | Formulations may be specific to ndPCR (e.g., QIAcuity Probe Master Mix) or ddPCR [65]. |
| Primers & Probes | Sequence-specific oligonucleotides for target amplification and detection. | Designed with the same stringency as for qPCR. Hydrolysis probes (e.g., FAM-labeled) are commonly used [43] [65]. |
| Nanoplates | Microfluidic plates containing thousands of integrated nanowells. | Specific to ndPCR systems (e.g., QIAcuity 26k 24-well Nanoplates). They are single-use consumables [65]. |
| Droplet Generation Cartridge & Oil | For creating the water-in-oil emulsion in ddPCR. | Specific to ddPCR systems (e.g., Bio-Rad DG32 cartridges and Droplet Generation Oil). Single-use consumables [6]. |
| PCR-Grade Water | Nuclease-free water for diluting samples and preparing reaction mixes. | A universal reagent critical for preventing enzymatic degradation of nucleic acids. |
| Restriction Enzymes | Used to digest complex DNA (e.g., genomic DNA) to improve access to the target sequence. | Can enhance precision, especially in ddPCR assays for targets like protists [8]. |
| Fluorescent Dyes/BHQ Probes | For detection. Proses are quenched (e.g., with BHQ-1) to reduce background fluorescence [43]. | Universal for probe-based detection. |
Cost-Benefit Analysis: Research vs. Regulated Environments
The choice between ddPCR and ndPCR is heavily influenced by the operational context, with distinct advantages emerging for research versus regulated environments like Quality Control (QC) in Good Manufacturing Practice (GMP) settings.
Table 3: Cost-Benefit Analysis for Research and Regulated Environments
| Factor | Droplet Digital PCR (ddPCR) | Nanoplate Digital PCR (ndPCR) |
|---|---|---|
| Upfront Instrument Cost | Varies; may require multiple modules (generator, cycler, reader). | Integrated instrument; potentially lower total cost of ownership. |
| Consumable Cost | Cost per sample for droplets/generation oil. | Cost per sample for nanoplates. |
| Hands-on Time / Labor | High (multiple instruments and transfer steps) [16] [6]. | Low (streamlined, "sample-in, results-out" workflow) [16] [29]. |
| Throughput & Scalability | High throughput options available in research settings [16]. | High throughput with 96-well plates; suited for batch processing. |
| Ease of Use & Training | Requires trained personnel; workflow is more complex [6]. | qPCR-like workflow; minimal training required; easy to adopt [63] [6]. |
| Multiplexing Capability | Limited in older models, newer systems can detect up to 12 targets [16]. | High multiplexing capability (e.g., up to 5-plex in a single well) [16] [6]. |
| Robustness & Contamination | Risk of droplet coalescence, shearing, and cross-contamination during transfers [6]. | Closed partitioning system minimizes contamination risk; highly robust [43] [6]. |
| Data Reproducibility | Can be affected by droplet variability and "rain" [6]. | High reproducibility due to fixed, uniform partition size and volume [6]. |
| GMP/Regulatory Compliance | Possible to validate, with extensive literature precedent [16]. | Ideal for QC. Integrated system with audit trail and 21 CFR Part 11 compliant software features [16]. |
| Ideal Application Context | Fundamental research, assay development, applications requiring ultra-high numbers of partitions [16]. | Routine QC testing, clinical diagnostics, manufacturing environments requiring high throughput and robustness [16] [3]. |
Analysis for Research Environments
In basic and applied research, ddPCR has been the established workhorse. Its key advantage has been the very high number of partitions, which can provide a wider dynamic range and potentially higher resolution for detecting rare events [6]. The flexibility to optimize droplet-based protocols and the extensive body of peer-reviewed literature supporting its use makes it a powerful tool for exploratory science, such as characterizing cells during process development in cell therapy [16]. The primary trade-offs are the more labor-intensive workflow and the need for technical expertise to generate high-quality, reproducible data.
Analysis for Regulated Environments
In regulated environments such as QC labs in cell and gene therapy manufacturing, ndPCR offers distinct and compelling advantages. The "sample-in, results-out" workflow drastically reduces hands-on time, minimizes the potential for human error, and lowers the risk of contamination—a critical factor for maintaining sample integrity in release assays [16]. The integrated nature of the instruments often includes features that support 21 CFR Part 11 compliance, such as security, auditing, and e-signature capabilities [16]. As noted by RoslinCT, for "critical QC release assays in cell and gene therapy manufacturing, factors such as convenience, robustness, and streamlined workflows are paramount," making ndPCR platforms their current workhorse for GMP manufacturing [16].
The decision between droplet digital PCR and nanoplate digital PCR is not a matter of declaring one technology universally superior. Rather, it is a strategic choice based on the specific needs of the application and the operational environment. ddPCR, with its high partition count and established history, remains a powerful and flexible tool for discovery-phase research and specialized applications. Conversely, ndPCR, with its streamlined workflow, reduced hands-on time, enhanced robustness, and integrated compliance features, presents a compelling and often more efficient solution for high-throughput, routine testing, and regulated environments like clinical diagnostics and quality control in drug development. As the field of molecular diagnostics advances, this fit-for-purpose approach will ensure that researchers and developers can leverage the full power of digital PCR with maximum efficiency and reliability.
Conclusion
The choice between nanoplate and droplet digital PCR is not a matter of one technology being universally superior, but rather a strategic decision based on application-specific needs. Recent 2025 studies consistently demonstrate that both platforms deliver high precision and accuracy, with performance often converging in well-optimized assays. Nanoplate systems excel with streamlined, automated workflows that reduce hands-on time and human error, making them highly suitable for quality control and clinical diagnostics. Droplet systems offer proven flexibility and a strong track record in research. Key differentiators often lie in workflow efficiency, ease of use, and integration into regulated environments, rather than fundamental quantification performance. Future developments will likely focus on increasing multiplexing capabilities, further automating workflows, and establishing standardized validation protocols to fully realize the potential of dPCR in personalized medicine and routine clinical testing.
References
- 1. Quantitative or digital PCR? A comparative analysis for ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993624001584]
- 2. Digital PCR: from early developments to its future application ... [https://pubs.rsc.org/en/content/articlehtml/2025/lc/d5lc00055f]
- 3. Comparative Performance of Digital PCR and Real-Time RT ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12474457/]
- 4. Comparison of digital PCR systems for the analysis ... [https://www.sciencedirect.com/science/article/pii/S0009898123000414]
- 5. Comparative Analysis of Two Digital PCR Platforms for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12361875/]
- 6. Comparison of digital PCR methods [https://www.qiagen.com/us/knowledge-and-support/knowledge-hub/bench-guide/pcr/digital-pcr/comparison-of-digital-pcr-methods]
- 7. Development of a Nanoplate-Based Digital PCR Test ... [https://www.idcmjournal.org/quantitative-detection-of-hadv-using-nanoplate-based-dpcr-system]
- 8. Comparing the precision of two digital PCR applications for ... [https://www.nature.com/articles/s41598-025-13143-8]
- 9. Comparative Performance of Digital PCR and Real-Time ... [https://www.mdpi.com/1999-4915/17/9/1259]
- 10. 【新品发布】DdPCR试剂盒开发与验证 [https://www.biomart.cn/news/16/2911612.htm]
- 11. 一种用于制备数字pcr微滴的油相组合物及其应用 [https://patents.google.com/patent/WO2020228257A1/zh]
- 12. DDPCR 检测_南京科佰生物科技有限公司 [https://www.cobioer.com/service/info7.html]
- 13. Performance Comparison of Droplet Digital PCR and Next ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12062871/]
- 14. Performance Comparison of Droplet Digital PCR and Next- ... [https://pubmed.ncbi.nlm.nih.gov/40346007/]
- 15. Digital PCR: from early developments to its future application ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12278255/]
- 16. dPCR vs. ddPCR: Why Precision Matters in Cell and Gene ... [https://www.roslinct.com/insights/dpcr-vs-ddpcr/]
- 17. Superior and secure partitioning in dPCR nanoplates [https://www.qiagen.com/us/product-categories/discovery-and-translational-research/pcr-qpcr-dpcr/dpcr-assays-kits-and-instruments/dpcr-accessories]
- 18. Digital PCR outperforms quantitative real-time PCR for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12288185/]
- 19. Duplex nanoplate-based digital PCR assay for ... [https://www.sciencedirect.com/science/article/abs/pii/S0889157525000602]
- 20. Comparison of two digital PCR platforms for quantification ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12622311/]
- 21. Comparing digital and real-time PCR platforms for ... [https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5788655]
- 22. First nanoplate digital PCR method to trace allergenic foods [https://www.sciencedirect.com/science/article/pii/S030881462400298X]
- 23. Nanoarray Digital Polymerase Chain Reaction with High- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8645066/]
- 24. Present and Future Applications of Digital PCR in ... [https://www.mdpi.com/2075-4418/14/9/931]
- 25. dPCR: A Technology Review - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC5948698/]
- 26. Digital droplet PCR is an accurate and precise method to ... [https://www.nature.com/articles/s41598-025-20944-4]
- 27. Evaluating the sensitivity of droplet digital PCR for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10903512/]
- 28. Comparing the precision of two digital PCR applications for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12297429/]
- 29. Digital PCR workflow and products [https://www.qiagen.com/us/applications/digital-pcr/workflow-and-products]
- 30. QIAcuity Digital PCR System [https://www.qiagen.com/us/products/instruments-and-automation/pcr-instruments/qiacuity-digital-pcr-system]
- 31. In-house validation of a droplet digital PCR system using a ... [https://www.sciencedirect.com/science/article/pii/S0003267025005665]
- 32. Assessment of the precision and sensitivity of QIAcuity ... [https://www.qiagen.com/us/resources/resourcedetail?id=c8a501e8-ebed-49f6-a47e-b2e7a6849c6e&lang=en]
- 33. dPCR LNA Mutation Assay (200) [https://www.qiagen.com/us/products/instruments-and-automation/accessories/dpcr-lna-mutation-assays]
- 34. Digital PCR-dPCR Market Insights & Growth Outlook 2025–2032 [https://www.congruencemarketinsights.com/report/digital-pcr-dpcr-market]
- 35. Droplet Digital PCR: A Powerful Tool for Accurate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12080313/]
- 36. Analytical and Clinical Performance of Droplet Digital PCR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8316104/]
- 37. Droplet digital PCR method for the absolute quantitative ... [https://www.sciencedirect.com/science/article/pii/S0740002023000527]
- 38. Development and validation of nanoplate-based RT-dPCR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12085003/]
- 39. Validation of Droplet Digital Polymerase Chain Reaction ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2020.01512/full]
- 40. Digital PCR as an effective tool for GMO quantification in ... [https://www.sciencedirect.com/science/article/pii/S0308814619308271]
- 41. Cell and Gene Therapy Manufacturing Using Digital PCR [https://www.thermofisher.com/us/en/home/life-science/pcr/digital-pcr/cell-gene-therapy-manufacturing.html]
- 42. A quality control revolution for droplet digital PCR [https://www.nature.com/articles/d42473-020-00123-x]
- 43. Development of a Nanoplate-Based Digital PCR Test ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10986707/]
- 44. Validation of a digital PCR method for quantification ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5007884/]
- 45. Development and validation of nanoplate -based RT-dPCR assay for... [https://bmcvetres.biomedcentral.com/articles/10.1186/s12917-025-04807-8]
- 46. Methylation-sensitive restriction enzyme-droplet digital ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10998716/]
- 47. Determining lower limits of detection of digital PCR assays ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5129438/]
- 48. strategies for Digital and... polymerase chain reaction accurate [https://pmc.ncbi.nlm.nih.gov/articles/PMC10123956/]
- 49. Quantitative Analysis of Food and Feed Samples with Droplet ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0062583]
- 50. A ddPCR method for simultaneous detection and ... [https://www.nature.com/articles/s41598-025-17272-y]
- 51. Multiplexed Digital PCR Reference Gene Measurement for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12523864/]
- 52. A Novel Digital PCR Assay for Accurate Detection and ... [https://www.mdpi.com/2072-6694/17/5/811]
- 53. Guide to multiplexing dPCR assays [https://www.qiagen.com/us/applications/digital-pcr/beginners/dpcr-multiplexing]
- 54. Digital PCR Multiplex Assay Optimization [https://www.stillatechnologies.com/digital-pcr/primer-design/multiplex-assay-optimization/]
- 55. the Comparing of two precision applications for copy... digital PCR [https://www.nature.com/articles/s41598-025-13143-8?error=cookies_not_supported&code=65bb69cc-66c5-45ae-bb59-6cd842ccfdfa]
- 56. Application of droplet for the detection of vector copy... digital PCR [https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-020-02358-0]
- 57. Medical diagnostic value of digital PCR (dPCR) [https://www.sciencedirect.com/science/article/pii/S2667099223000221]
- 58. Droplet digital PCR of viral DNA/RNA, current progress ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8013307/]
- 59. Digital PCR Applications | Thermo Fisher Scientific - ES [https://www.thermofisher.com/us/en/home/life-science/pcr/digital-pcr/applications.html]
- 60. What Are the Advantages of ddPCR Over Other PCR ... [https://mogene.com/what-are-the-advantages-of-ddpcr-over-other-pcr-variants/]
- 61. Applications of PCR in Drug Development [https://kcasbio.com/blogs/pcr-applications-drug-development/]
- 62. Droplet digital PCR analysis of CDH13 methylation status ... [https://www.nature.com/articles/s41598-024-65580-6]
- 63. Digital PCR | dPCR technology [https://www.qiagen.com/us/applications/digital-pcr]
- 64. A low-cost, programmable, and multi-functional droplet ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400521012466]
- 65. Development and Application of Reverse Transcription ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11309843/]