Enhancing Precision in Digital PCR: The Critical Role of Restriction Enzymes
This article provides a comprehensive analysis of the impact of restriction enzymes on the precision and accuracy of digital PCR (dPCR) for researchers and drug development professionals.
Enhancing Precision in Digital PCR: The Critical Role of Restriction Enzymes
Abstract
This article provides a comprehensive analysis of the impact of restriction enzymes on the precision and accuracy of digital PCR (dPCR) for researchers and drug development professionals. It explores the foundational principle of how enzymatic digestion enhances DNA target accessibility, details established and emerging methodological protocols, offers evidence-based strategies for troubleshooting and optimizing reactions, and presents rigorous cross-platform validation data. By synthesizing findings from recent comparative studies and clinical applications, this review serves as a definitive guide for implementing restriction enzyme-digested dPCR to achieve robust, reproducible nucleic acid quantification in complex genomic analyses, including copy number variation and methylation studies.
Why Digestion Matters: Unlocking DNA Accessibility in Digital PCR
Digital PCR (dPCR) is a powerful method for the absolute quantification of target nucleic acids that differs fundamentally from quantitative real-time PCR (qPCR). While qPCR relies on calibration curves and monitors amplification throughout the thermal cycling process, dPCR partitions samples into thousands of independent reactions, detects amplified targets via end-point measurement, and uses Poisson statistics to determine target concentration without external calibration [1] [2]. This partitioning approach allows dPCR to distinguish between merely present targets and those that are truly amplifiable, as only accessible targets will generate positive signals in their partitions after PCR amplification [1] [3]. The core principle of target accessibility states that dPCR specifically quantifies template molecules that can be successfully amplified under the given reaction conditions, making it particularly valuable for applications requiring high precision, such as copy number variation analysis, rare mutation detection, and environmental monitoring [4] [5].
FAQs on dPCR and Target Accessibility
How does dPCR specifically measure amplifiable rather than just present targets?
dPCR measures amplifiable targets through its partitioning approach and end-point detection. When a sample is partitioned into thousands of individual reactions, each partition functions as a separate PCR microreactor. After complete PCR amplification, only partitions containing targets that were accessible to primers and polymerase and successfully amplified will fluoresce as positive [1] [2]. Targets that are present but not amplifiable due to damage, secondary structure, or bound inhibitors will not generate amplification and thus will not be counted as positive events. This binary detection system (0 for no amplification, 1 for successful amplification) specifically enumerates molecules capable of amplification under the reaction conditions [1].
Why does template quality affect dPCR results more significantly than qPCR in some cases?
Template quality disproportionately affects dPCR because the technique relies on single-molecule amplification events in partitioned reactions. Unlike qPCR, which monitors amplification kinetics in a bulk reaction where partial template degradation might be compensated by efficient amplification of intact templates, dPCR requires each target molecule to be independently amplifiable [6] [7]. Damaged or inaccessible templates in individual partitions will fail to amplify, leading to underestimation of concentration. Template issues including poor integrity, nicking, residual PCR inhibitors, or complex secondary structures can all prevent amplification at the single-molecule level [6] [8].
How do restriction enzymes improve target accessibility in dPCR?
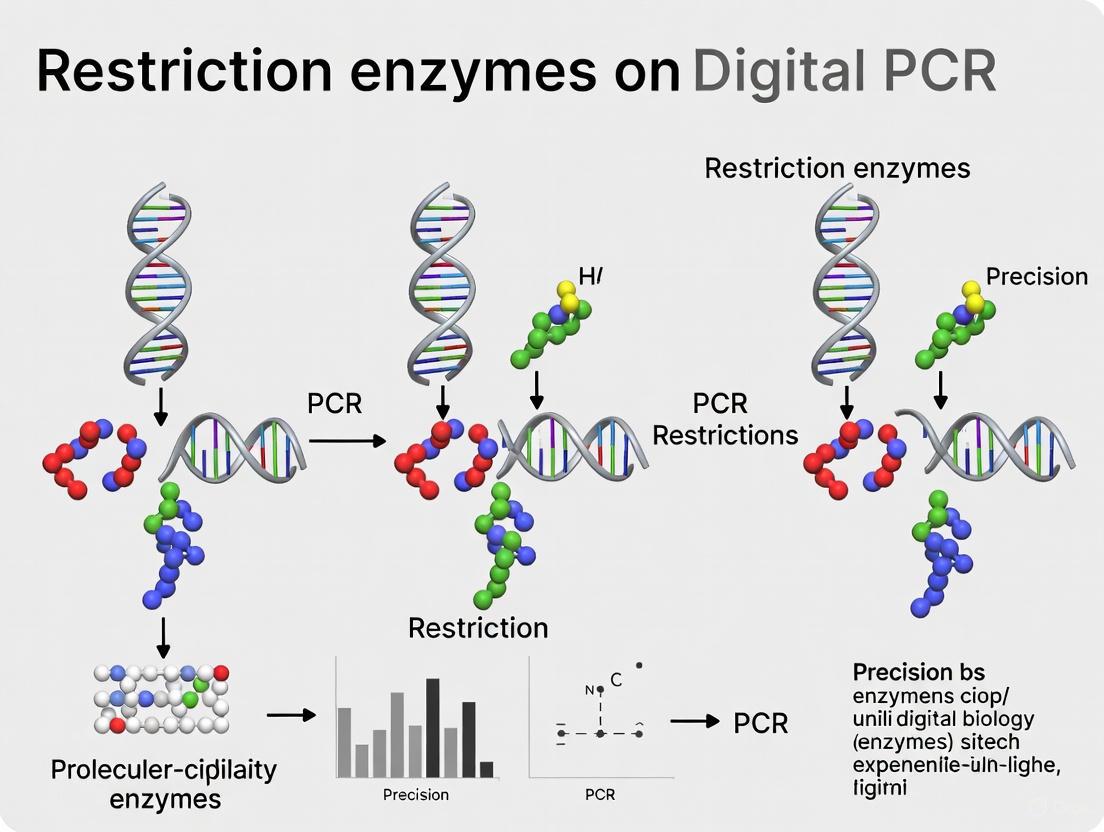
Restriction enzymes significantly enhance target accessibility, particularly for complex templates or tandemly repeated genes, by cutting DNA at specific recognition sites. This process helps to: (1) separate target sequences from surrounding genomic DNA that might impede primer access, (2) resolve secondary structures that prevent efficient amplification, and (3) linearize circular templates for better primer binding [4] [3]. Research has demonstrated that restriction enzyme selection directly impacts measurement precision, with different enzymes yielding varying results due to their specific cutting patterns and efficiency [4].
Table 1: Impact of Restriction Enzyme Selection on dPCR Precision
| Platform | Restriction Enzyme | Precision (CV Range) | Key Findings |
|---|---|---|---|
| QX200 ddPCR | EcoRI | 2.5% - 62.1% | High variability, especially at lower template concentrations |
| QX200 ddPCR | HaeIII | <5% for all concentrations | Greatly improved precision across all template levels |
| QIAcuity One ndPCR | EcoRI | 0.6% - 27.7% | Moderate variability, better performance than ddPCR with same enzyme |
| QIAcuity One ndPCR | HaeIII | 1.6% - 14.6% | Good precision with less dramatic improvement than ddPCR |
What factors determine whether a target is "amplifiable" in dPCR?
A target is considered amplifiable in dPCR when it meets several criteria: (1) it must be structurally intact at the primer binding sites and amplification region, (2) it must be free of bound proteins or inhibitors that would prevent polymerase access, (3) it must not possess secondary structures that block polymerase progression, (4) it must be in a physical state that allows primer annealing (e.g., linearized rather than highly supercoiled), and (5) it must be present in a partition with all necessary reaction components [6] [7] [8]. The numerous chemical and physical barriers that can prevent amplification highlight why dPCR typically reports lower concentrations than methods that merely detect presence of DNA sequences.
Troubleshooting Guide: Addressing Target Accessibility Issues in dPCR
Table 2: Troubleshooting Common Target Accessibility Problems in dPCR
| Problem | Possible Causes | Recommended Solutions |
|---|---|---|
| Low precision between replicates | Inefficient restriction enzyme digestion, template secondary structures | Test different restriction enzymes (e.g., HaeIII instead of EcoRI), add digestion optimization step [4] |
| Lower than expected copy numbers | Template damage, PCR inhibitors, secondary structures | Repurify template DNA, use DNA repair mix, include GC enhancers for difficult templates [6] [8] |
| Partition saturation at high concentrations | Too much input DNA, insufficient partitioning | Dilute sample appropriately, ensure optimal partition number for expected concentration [1] |
| Non-specific amplification | Primer dimers, mispriming | Optimize primer design, use hot-start polymerases, increase annealing temperature [7] [8] |
| Smearing or high background | Contamination, overcycling, poor primer specificity | Establish separate pre- and post-PCR areas, use aerosol filter tips, reduce cycle number [7] |
Experimental Protocols for Assessing Target Accessibility
Protocol 1: Restriction Enzyme Optimization for Improved Precision
This protocol evaluates how different restriction enzymes affect measurement precision in dPCR, particularly for targets with potential accessibility issues [4].
Sample Preparation: Select DNA samples representing varying concentrations of your target, including both high-copy and low-copy samples if possible.
Restriction Enzyme Selection: Choose at least two restriction enzymes with different recognition sites. Include one enzyme that cuts near your target region and one that cuts farther away if sequence information is available.
Digestion Reaction Setup:
- Prepare separate digestion reactions for each enzyme:
- 1μg genomic DNA
- 1X restriction enzyme buffer
- 10 units restriction enzyme (e.g., EcoRI, HaeIII)
- Nuclease-free water to 20μL
- Incubate at enzyme-specific temperature (typically 37°C) for 1 hour
- Heat-inactivate enzymes according to manufacturer instructions
- Prepare separate digestion reactions for each enzyme:
dPCR Setup:
- Prepare dPCR reactions using digested templates
- Use consistent primer/probe concentrations across all samples
- Include undigested control samples for comparison
Analysis:
- Calculate coefficient of variation (CV) for replicates across enzyme conditions
- Compare measured copy numbers between different digestion treatments
- Assess signal intensity and separation between positive and negative partitions
Protocol 2: Assessing Amplifiability Through Spike-In Controls
This protocol uses internal controls to distinguish between template presence and amplifiability.
Control Design: Select or design a control template that is similar to your target but contains a different probe-binding region for multiplex detection.
Sample Processing:
- Divide sample into two aliquots
- Treat one aliquot with DNA repair mix (e.g., PreCR Repair Mix) according to manufacturer instructions
- Leave second aliquot untreated
dPCR Reaction Setup:
- Set up multiplex dPCR reactions containing:
- Primers/probes for your target
- Primers/probes for the control template
- Treated and untreated sample aliquots
- Use appropriate positive and negative controls
- Set up multiplex dPCR reactions containing:
Data Interpretation:
- Compare target concentration in repaired vs. unrepaired samples
- Significant increases after repair indicate presence of damaged templates
- Calculate ratio of amplifiable to total targets based on control measurements
Research Reagent Solutions for Target Accessibility
Table 3: Essential Reagents for Optimizing Target Accessibility in dPCR
| Reagent/Category | Function in Improving Accessibility | Examples/Specific Recommendations |
|---|---|---|
| Restriction Enzymes | Linearize DNA, resolve secondary structures, improve primer access | HaeIII (showed superior precision in studies), enzyme with recognition sites near target region [4] |
| DNA Polymerase | Efficient amplification of single molecules, tolerance to inhibitors | Hot-start polymerases, high-processivity enzymes for complex templates [6] [8] |
| PCR Additives/Co-solvents | Reduce secondary structure, improve efficiency for difficult templates | GC enhancers, DMSO, betaine for GC-rich templates [6] [7] |
| DNA Repair Mixes | Restore amplifiability to damaged templates | PreCR Repair Mix for repairing nicked, oxidized, or damaged DNA [8] |
| Purification Kits | Remove inhibitors, improve template quality | Silica membrane-based kits, magnetic bead systems for clean template isolation [6] [7] |
Visualization of dPCR Workflow and Target Accessibility
The principle of target accessibility underscores that dPCR specifically quantifies amplifiable—not just present—nucleic acid targets. This distinction is crucial for applications requiring high precision, such as clinical diagnostics and environmental monitoring. Through strategic experimental design, including restriction enzyme optimization and careful template preparation, researchers can significantly improve dPCR accuracy and reliability. The protocols and troubleshooting guidance provided here offer practical approaches to address target accessibility challenges, enabling researchers to obtain more meaningful and reproducible results from their dPCR experiments.
Frequently Asked Questions (FAQs)
Q1: Why is long, complex genomic DNA particularly challenging for digital PCR quantification?
Long, complex genomic DNA presents two main challenges for accurate digital PCR (dPCR) quantification. First, high-molecular-weight templates with complex structures can lead to uneven partitioning during the dPCR process. If DNA molecules are too large, they may not partition randomly into the reaction chambers (nanoplates or droplets) as assumed by the Poisson statistics, potentially causing over-quantification [9]. Second, if the target gene exists in tandem repeats or linked gene copies on the same DNA molecule, a single positive partition may contain multiple target copies. dPCR would count this as a single positive event, leading to an under-estimation of the true copy number [9].
Q2: How does the use of a restriction enzyme improve the accuracy of dPCR for complex DNA?
Restriction enzymes digest long, complex genomic DNA into smaller fragments, which addresses the core physical barriers to quantification [9]. This digestion provides several key benefits:
- Enhances Partitioning: It reduces sample viscosity, allowing for more accurate pipetting and uniform distribution of DNA molecules across thousands of partitions [9].
- Separates Linked Copies: It physically severs the linkage between tandemly repeated gene copies, ensuring that each copy can independently segregate into a partition. This allows each copy to be counted individually, leading to accurate absolute quantification [10] [11].
- Linearizes Plasmid DNA: For supercoiled plasmids, restriction digestion linearizes the DNA, improving the efficiency of primer and probe binding [9].
Q3: What are the critical factors to consider when selecting a restriction enzyme for a dPCR assay?
The most critical factor is that the restriction enzyme must not cut within the amplicon sequence defined by your primers and probe [9]. If it does, the target sequence will be destroyed, and no amplification will occur. Beyond this, selection can be based on the recognition site. The table below lists enzymes commonly recommended for dPCR.
| Restriction Enzyme | Recognition Site | Notes |
|---|---|---|
| HaeIII [4] [10] | GG/CC | Used in a comparative platform study; recommended by Bio-Rad. |
| AluI [10] | AG/CT | Recommended by Bio-Rad. |
| MseI [10] | T/TAA | Recommended by Bio-Rad. |
| EcoRI [4] [10] | G/AATTC | Used in a comparative platform study. |
| HinfI [10] | G/ANTC | Available for dPCR use. |
Q4: What is the evidence that restriction enzyme choice impacts measurement precision?
Recent research directly comparing the precision of different dPCR platforms found that the choice of restriction enzyme significantly affected results. A 2025 study showed that using HaeIII instead of EcoRI substantially increased precision, especially for the QX200 droplet digital PCR (ddPCR) system. For ddPCR, the coefficient of variation (CV) using EcoRI varied widely (2.5% to 62.1%), but all CVs fell below 5% when using HaeIII [4]. This demonstrates that enzyme selection is a key variable for obtaining robust, reproducible data.
Troubleshooting Guide
Problem: Inconsistent or Imprecise Copy Number Results
- Potential Cause 1: Non-uniform DNA partitioning due to large fragment size.
- Solution: Digest the genomic DNA with a restriction enzyme prior to the dPCR run. Use 10 units of enzyme per microgram of DNA and incubate for 5-60 minutes. No cleanup is necessary before adding the sample to the dPCR reaction [10].
- Potential Cause 2: The selected restriction enzyme cuts within the amplicon.
- Solution: Carefully check your primer and probe sequences against the recognition site of your chosen enzyme. Re-design your assay or select a different enzyme that does not cut the target amplicon [9].
- Potential Cause 3: The DNA input amount is too high, leading to over-saturation of partitions.
- Solution: Ensure the average number of target copies per partition is between 0.5 and 3 for optimal quantification [9]. Calculate the required DNA input based on your genome size. For example, 10 ng of human gDNA (haploid genome size ~3.3 billion bp) contains approximately 3,000 copies of a single-copy gene [9].
Problem: Failed or Inefficient Droplet Generation
- Potential Cause: High viscosity of the sample due to long, intact genomic DNA.
- Solution: Perform an in-line restriction digest. Assemble the dPCR reaction at room temperature and add 0.5–1 µL of restriction enzyme (5–20 units) directly to the reaction mix. The enzyme will digest the DNA during setup and be inactivated during the first PCR denaturation step [10].
Experimental Protocol: Restriction Enzyme Digestion for dPCR
This protocol can be performed as a separate step or directly in the dPCR reaction mix.
- Assemble Reaction: Prepare the dPCR reaction mix at room temperature as you would for a standard assay.
- Add Enzyme: Add 0.5–1 µL of each restriction enzyme (providing 5–20 units) to the reaction mixture.
- Proceed with Workflow: After setup, continue directly with droplet or nanoplate generation according to your instrument's protocol. The restriction enzyme will be active during sample preparation and inactivated during the first high-temperature denaturation step of the PCR cycle.
- Set-up Digest: Assemble the restriction digest using the manufacturer's recommended buffer.
- Use Recommended Amount: Use 10 units of restriction enzyme per microgram of DNA sample.
- Incubate: Incubate for 5–60 minutes at the enzyme's optimal reaction temperature.
- Optional Inactivation: Heat inactivation is optional but can be performed.
- No Cleanup: It is not necessary to clean up the digest reaction. The sample can be directly added to the dPCR master mix, but avoid carrying over more than 1/10 of the total reaction volume from the restriction digest mixture.
The following table summarizes key quantitative findings from a recent 2025 study that compared the performance of different dPCR platforms and the impact of restriction enzymes [4].
Table 1: Platform Comparison and the Impact of Restriction Enzymes on dPCR Precision [4]
| Parameter / Finding | QIAcuity One Nanoplate dPCR (ndPCR) | QX200 Droplet Digital PCR (ddPCR) |
|---|---|---|
| Limit of Detection (LOD) | ~0.39 copies/µL input | ~0.17 copies/µL input |
| Limit of Quantification (LOQ) | ~1.35 copies/µL input | ~4.26 copies/µL input |
| Precision with EcoRI | CV range: 0.6% - 27.7% | CV range: 2.5% - 62.1% |
| Precision with HaeIII | CV range: 1.6% - 14.6% | CV range: < 5% (all samples) |
| Key Conclusion on Enzymes | Enzyme choice had less impact on overall precision. | Precision was dramatically improved using HaeIII instead of EcoRI. |
Workflow Visualization
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for dPCR of Complex Genomic DNA
| Item | Function in the Protocol |
|---|---|
| Restriction Enzymes (e.g., HaeIII, AluI) | Digests long genomic DNA to reduce viscosity and separate tandemly repeated gene copies, ensuring accurate and precise quantification [4] [10]. |
| Digital PCR Master Mix | A specialized buffer containing DNA polymerase, dNTPs, and optimized salts. The choice of master mix can be a critical factor for the accuracy of the system [3]. |
| Sequence-Specific Primers & Probes | Binds to the target DNA sequence for amplification and detection. Higher concentrations than in qPCR are often used in dPCR to increase fluorescence intensity and improve cluster separation [9]. |
| Nuclease-Free TE Buffer (pH 8.0) | Recommended for resuspending and storing lyophilized primers and probes to ensure their stability and prevent degradation. Probes with Cy5/Cy5.5 should be stored in TE Buffer, pH 7.0 [9]. |
| Positive & Negative Controls | Validates the performance of the assay. A positive control confirms amplification, while a negative control (NTC) monitors for contamination [9]. |
Scientific Rationale: Why Use Restriction Enzymes in Digital PCR?
What is the core principle behind using restriction enzymes to improve dPCR?
Digital PCR (dPCR) achieves absolute quantification by partitioning a sample into thousands of reactions and counting positive amplifications. However, it does not estimate the absolute number of DNA targets in a volume, but rather the number of accessible and amplifiable targets [12]. Intact genomic DNA, with its complex and folded structure, can have target sequences that are physically inaccessible to PCR primers and polymerase. This can lead to an underestimation of the true copy number.
How do restriction enzymes solve this problem?
Restriction enzymes work as molecular scissors that cleave DNA at specific recognition sites. This enzymatic fragmentation performs a crucial pretreatment step [12]:
- Unfolds DNA Structure: By cutting long DNA strands into smaller fragments, restriction enzymes release tightly bound DNA, making target sequences more accessible.
- Enhances Target Availability: This increased accessibility can improve the efficiency of the subsequent PCR, ensuring that a higher proportion of the true target molecules are amplified and detected.
The diagram below illustrates this core principle and its effect on dPCR precision.
Troubleshooting Guide: Common Restriction Enzyme Digestion Problems
Even with a sound principle, experimental outcomes can vary. The table below outlines common issues, their causes, and solutions to ensure successful enzymatic pretreatment for dPCR [13] [14].
| Problem | Possible Cause | Recommended Solution |
|---|---|---|
| Incomplete or No Digestion | Inactive enzyme, suboptimal buffer, DNA contaminants, methylation, excess glycerol | Verify enzyme storage conditions (-20°C, minimize freeze-thaw). Use manufacturer's recommended buffer. Repurify DNA to remove inhibitors (e.g., salts, SDS, EDTA). Check for methylation sensitivity; use dam-/dcm- E. coli strains or methylation-insensitive enzymes. Keep final glycerol concentration <5% [13] [14]. |
| Unexpected Cleavage Pattern | Star activity (off-target cleavage), contamination with another enzyme, unexpected DNA sequences | Reduce enzyme units (≤10 U/μg DNA). Avoid prolonged incubation. Use recommended salt/pH conditions. Prepare fresh enzyme/buffer stocks. Verify DNA template sequence and cloning strategy [13] [14]. |
| Diffuse or Smeared DNA Bands | Poor DNA quality (degraded), nuclease contamination in reagents | Run undigested DNA on gel; if smearing is present, repurify DNA. Use fresh, molecular biology-grade reagents and nuclease-free water [13]. |
Experimental Protocols: Key Methodologies from Cited Research
Protocol: Evaluating Restriction Enzymes for dPCR Precision
This protocol is adapted from a study comparing the precision of nanoplate-based (ndPCR) and droplet-based (ddPCR) digital PCR systems [4].
- 1. DNA Sample Preparation: Use DNA extracted from a known number of cells (e.g., the ciliate Paramecium tetraurelia) or synthetic oligonucleotides.
- 2. Restriction Enzyme Digestion:
- Set up digestion reactions with your DNA sample.
- Test different restriction enzymes (e.g., HaeIII and EcoRI were used in the study [4]).
- Use the manufacturer's recommended buffer and incubation temperature (typically 37°C).
- Use 3-5 units of enzyme per µg of DNA and incubate for 1 hour.
- Include a control without restriction enzyme.
- 3. Digital PCR Setup:
- Prepare dPCR reactions using the digested and undigested control DNA.
- Use a platform such as the QIAcuity One (nanoplate-based) or QX200 (droplet-based) [4].
- Follow the platform-specific protocol for partition generation and thermocycling.
- 4. Data Analysis:
- Calculate the estimated gene copy number and precision (Coefficient of Variation, %CV) for each sample and enzyme condition.
- Compare the results to determine which restriction enzyme provides the highest precision and best agreement with expected cell counts.
Protocol: dPCR for Human Genomic DNA Quantification
This protocol is based on research that used dPCR to value-assign human genomic DNA reference materials [12].
- 1. Sample and Enzyme Selection:
- Select human genomic DNA samples from characterized sources.
- Choose multiple, well-validated PCR assays targeting single-copy genes.
- Test a panel of four restriction enzymes (e.g., as done with chamber dPCR and droplet dPCR platforms [12]).
- 2. Parallel Digestion and dPCR:
- Perform restriction digests on aliquots of the same DNA sample with each enzyme separately.
- Also include an "uncut" sample for comparison.
- Run all digested and undigested samples on the chosen dPCR platform(s).
- 3. Analysis and Interpretation:
- For each PCR assay, compare the estimated copy number concentration obtained from cut vs. uncut DNA.
- Note that while restriction can increase accessibility, it can also reduce the number of amplifiable targets if a cut occurs within the PCR amplicon. Therefore, the goal is to find an enzyme that fragments the genome without disrupting your specific target sequences [12].
The following table summarizes quantitative findings from a comparative study, highlighting how enzyme choice directly impacts experimental precision [4].
Table: Precision (Coefficient of Variation, %CV) with Different Restriction Enzymes
| Number of Cells | ndPCR with EcoRI | ndPCR with HaeIII | ddPCR with EcoRI | ddPCR with HaeIII |
|---|---|---|---|---|
| 5 Cells | 27.7% | 14.6% | 62.1% | <5% |
| 10 Cells | 1.8% | 1.6% | 10.2% | <5% |
| 50 Cells | 0.6% | 2.3% | 2.5% | <5% |
| 100 Cells | 2.0% | 3.0% | 5.7% | <5% |
Key Conclusion: The data demonstrates that HaeIII significantly improved precision, especially for the ddPCR system, where it reduced CV from a highly variable range (2.5%-62.1%) to a consistently low value (under 5%) across all cell numbers tested [4].
The Scientist's Toolkit: Essential Research Reagents
| Item | Function in the Experiment |
|---|---|
| Restriction Enzymes (e.g., HaeIII, EcoRI) | Enzymatically fragment genomic DNA to enhance target accessibility for PCR primers and polymerase [4] [12]. |
| Digital PCR System | Platform (e.g., nanoplate or droplet-based) that partitions samples to allow absolute quantification of nucleic acids without a standard curve [4] [15]. |
| Manufacturer's Reaction Buffer | Provides optimal salt and pH conditions to ensure maximum restriction enzyme activity and prevent star activity [13] [14]. |
| dam-/dcm- E. coli Strains | Host strains for propagating plasmid DNA to avoid methylation that could block cleavage by methylation-sensitive restriction enzymes [13] [14]. |
| Molecular Biology-Grade Water | Nuclease-free water used to prepare reaction mixes, preventing enzyme degradation and contamination [13]. |
Frequently Asked Questions (FAQs)
Q1: Why is my dPCR copy number estimate lower after adding a restriction enzyme? This can occur if the restriction enzyme cuts within the PCR amplicon itself, destroying the target sequence. Re-check the location of the enzyme's recognition sites relative to your primer binding sites. Choose an enzyme that does not cut within your amplicon [12].
Q2: How do I select the best restriction enzyme for my dPCR assay? The ideal enzyme should not cut within your target amplicon. If the sequence is known, perform an in silico digest. Enzymes like HaeIII have been shown empirically to improve precision in complex genomic DNA [4]. Testing a small panel of enzymes in a pilot experiment is highly recommended.
Q3: My restriction digest seems complete, but dPCR precision is still poor. What else should I check? First, ensure your dPCR reaction is in the "digital range" (sufficiently diluted so some partitions contain no template) [16]. Re-check DNA quality and concentration. Also, verify that the master mix and thermocycling conditions are optimized for your specific dPCR platform.
Q4: Can restriction enzymes be used with any dPCR chemistry? Yes, the principle is platform-agnostic. However, the degree of improvement may vary between systems (e.g., ddPCR vs. ndPCR) as shown in research [4]. Always follow the specific protocol for your dPCR platform when incorporating a digestion step.
Accurately quantifying gene copy numbers in environmental samples is fundamental to understanding microbial community dynamics and ecosystem functioning. Digital PCR (dPCR) has emerged as a powerful tool for absolute quantification of nucleic acids, offering superior sensitivity and precision compared to quantitative real-time PCR (qPCR) [4] [17]. However, even this advanced technology faces challenges when analyzing organisms with complex genomic architectures, particularly those with high or variable gene copy numbers.
This case study examines a critical methodological challenge encountered during gene copy number analysis of the ciliate Paramecium tetraurelia and demonstrates how strategic restriction enzyme selection rescued experimental precision. Ciliates present a particular quantification challenge because they can exhibit substantial gene copy number variations, ranging from a few thousand to half a million copies, with some genes occurring in tandem repeats that limit enzyme accessibility [4]. When researchers compared the performance of two digital PCR platforms - the QX200 droplet digital PCR (ddPCR) from Bio-Rad and the QIAcuity One nanoplate-based digital PCR (ndPCR) from QIAGEN - they made a crucial discovery: restriction enzyme choice significantly impacted measurement precision, especially for the droplet-based system [4].
Experimental Investigation: Quantifying the Restriction Enzyme Effect
Research Objective and Methodological Framework
The study aimed to compare the precision and accuracy of two dPCR platforms for copy number quantification in protists, using both synthetic oligonucleotides and DNA extracted from varying cell numbers of Paramecium tetraurelia [4]. A key component of the experimental design involved testing how different restriction enzymes affect gene copy number quantification accuracy and precision.
Experimental Protocol:
- Biological Material: DNA was extracted from precisely counted cell numbers of the ciliate Paramecium tetraurelia [4]
- Platform Comparison: QX200 ddPCR (Bio-Rad) vs. QIAcuity One ndPCR (QIAGEN) [4]
- Enzyme Comparison: Parallel tests with EcoRI and HaeIII restriction enzymes [4]
- Partitioning Characteristics:
- Precision Measurement: Coefficient of variation (CV%) across technical replicates was used to quantify precision [4]
Key Findings: Restriction Enzymes Dramatically Improved Precision
The quantitative results demonstrated a striking enzyme-dependent effect on measurement precision, particularly for the ddPCR platform.
Table 1: Impact of Restriction Enzyme Selection on Measurement Precision (CV%)
| Cell Numbers | ddPCR with EcoRI | ddPCR with HaeIII | ndPCR with EcoRI | ndPCR with HaeIII |
|---|---|---|---|---|
| 50 cells | 62.1% | <5% | 27.7% | 14.6% |
| 100 cells | 2.5% | <5% | 0.6% | 1.6% |
| Overall Range | 2.5-62.1% | <5% | 0.6-27.7% | 1.6-14.6% |
The data revealed that HaeIII consistently provided superior precision compared to EcoRI, with this effect being particularly dramatic for the ddPCR system [4]. When using EcoRI, the ddPCR platform showed unacceptably high variability (CV up to 62.1%), especially at lower cell counts [4]. However, when switching to HaeIII, precision improved dramatically, with all CV values below 5% for ddPCR [4]. While the ndPCR system showed less enzyme-dependent variation, HaeIII still provided improved precision, particularly at lower template concentrations [4].
Technical Guide: Restriction Enzyme Fundamentals for dPCR Applications
Core Function of Restriction Enzymes in dPCR
Restriction enzymes serve two critical functions in digital PCR applications:
- DNA Complexity Reduction: Large genomes like those of ciliates can interfere with droplet generation. Restriction digestion breaks DNA into manageable fragments, ensuring consistent partitioning [18].
- Tandem Repeat Resolution: For accurate copy number quantification, especially for genes in tandem repeats, restriction enzymes separate linked copies into discrete molecules, preventing undercounting [18].
Research Reagent Solutions
Table 2: Essential Reagents for Restriction Enzyme-dPCR Workflows
| Reagent Category | Specific Examples | Function & Importance |
|---|---|---|
| Restriction Enzymes | HaeIII, EcoRI, PvuII [4] [17] | Digest genomic DNA to resolve tandem repeats and reduce complexity |
| Reaction Buffers | Manufacturer-specific buffers [13] [19] | Provide optimal salt conditions and cofactors (Mg²⁺, DTT) for enzyme activity |
| DNA Purification Kits | QIAamp DNA Mini Kit [17] | Remove contaminants (SDS, EDTA, proteins) that inhibit restriction enzymes |
| dPCR Master Mixes | QIAcuity Probe PCR Kit [17] | Provide optimized reagents for partitioning and amplification |
| Nuclease-free Water | Molecular biology grade [13] [14] | Prevent enzyme degradation and nuclease contamination |
Troubleshooting Guide: Restriction Enzyme Issues in dPCR
Problem: Incomplete Digestion Leading to Poor Precision
Symptoms: High coefficient of variation (CV%) between replicates, underestimation of true copy number, inconsistent results across samples [4] [13].
Solutions:
- Enzyme Quality Control: Verify enzyme storage at -20°C, minimize freeze-thaw cycles (<3 cycles), check expiration dates [13] [14]
- Optimal DNA Concentration: Maintain DNA concentration between 20-100 ng/μL in the final reaction mixture [13] [19]
- Glycerol Concentration: Keep final glycerol concentration <5% in reaction mixture to prevent star activity [13] [19]
- Reaction Assembly: Add restriction enzyme last to the reaction mix and mix gently by pipetting (avoid vortexing) [14] [19]
- Incubation Time: Extend incubation time (1-2 hours) if digestion is incomplete [14] [20]
Problem: Unexpected Cleavage Patterns
Symptoms: Additional bands in gel electrophoresis, off-target cleavage, inaccurate fragment sizes [13] [19].
Solutions:
- Prevent Star Activity: Use recommended enzyme units (3-5 units/μg DNA), avoid prolonged incubation, ensure correct buffer conditions [19] [20]
- Check for Contamination: Use fresh enzyme and buffer stocks to avoid cross-contamination between different enzymes [19]
- Methylation Considerations: Use dam-/dcm- E. coli strains for plasmid propagation or select methylation-insensitive isoschizomers [13] [19]
- Verify Recognition Sites: Confirm restriction sites are present and accessible in template DNA; add flanking bases (4-8) for sites near DNA ends [13] [19]
Problem: Diffuse DNA Bands or Smearing
Symptoms: Poorly separated bands in gel electrophoresis, blurry or indistinct bands, difficulty interpreting results [13] [20].
Solutions:
- DNA Quality Assessment: Run undigested DNA on a gel; if smearing occurs, repurify DNA to remove nucleases [13] [20]
- Remove Enzyme Interference: Add 0.1-0.5% SDS to loading buffer and heat at 65°C for 10 minutes before electrophoresis to dissociate bound enzyme [19] [20]
- Nuclease Contamination: Replace reagents and use fresh enzyme stocks if nuclease contamination is suspected [20]
FAQs: Restriction Enzymes in Digital PCR
Q1: Why does restriction enzyme selection affect precision differently across dPCR platforms? A: The droplet-based ddPCR system appears more sensitive to DNA fragment size and distribution uniformity compared to nanoplate-based systems. HaeIII may generate more uniform fragment sizes that partition more consistently in droplets, explaining why it rescued precision specifically in the ddPCR platform [4].
Q2: How much restriction enzyme should I use in dPCR reactions? A: Use 3-5 units of enzyme per μg of DNA, but ensure the enzyme volume doesn't exceed 10% of the total reaction volume to maintain glycerol concentration below 5% [14] [19]. For challenging substrates like supercoiled plasmids, increase to 5-10 units/μg DNA [13].
Q3: Can restriction enzymes be used in multiplex dPCR applications? A: Yes, restriction enzymes are successfully used in multiplex dPCR. For example, one periodontal study used Anza 52 PvuII in a multiplex assay detecting three bacterial pathogens simultaneously [17]. Choose enzymes that work in a single buffer system for multiplex applications.
Q4: How does DNA methylation affect restriction enzyme efficiency in dPCR? A: Methylation can completely block some restriction enzymes from cutting their recognition sites. If working with bacterial DNA, consider DAM/DCM methylation. For eukaryotic DNA, CpG methylation may be an issue. Use methylation-insensitive enzymes or propagate plasmids in dam-/dcm- E. coli strains [13] [19].
Q5: What is the optimal order for setting up restriction digestion before dPCR? A: Use this recommended order: nuclease-free water → reaction buffer → DNA template → restriction enzyme (added last). This prevents enzyme exposure to concentrated buffer components that might cause premature inactivation [14] [19].
The case study demonstrates that restriction enzyme selection is not merely a technical step but a critical methodological factor that can determine experimental success in gene copy number analysis. Based on the findings:
- Platform-Specific Optimization is Essential: Restriction enzymes affect ddPCR and ndPCR platforms differently; optimize enzyme selection for your specific system [4].
- HaeIII Shows Superior Performance: For gene copy number studies, particularly with protists, HaeIII provided significantly better precision than EcoRI, especially for droplet-based systems [4].
- Systematic Troubleshooting Pays Off: When precision problems occur, follow methodical troubleshooting focusing on enzyme quality, reaction conditions, and DNA quality [13] [14].
- Quality Control is Non-Negotiable: Always include control digests with known DNA to verify enzyme activity and monitor for star activity or incomplete digestion [19].
The integration of appropriate restriction enzymes into dPCR workflows enables researchers to achieve the high precision required for accurate gene copy number analysis, even for challenging organisms like ciliates with complex genome structures. This approach has broad applications in environmental monitoring, clinical diagnostics, and fundamental biological research where precise nucleic acid quantification is essential [4] [17] [21].
From Theory to Bench: Protocols and Applications for Restriction Enzyme-dPCR
This guide addresses the integration of restriction enzymes directly into digital PCR (dPCR) reaction mixes. This one-tube workflow aims to streamline processes in precision oncology, antimicrobial resistance surveillance, and biopharmaceutical development by reducing handling steps and potential contamination. However, combining these enzymatic steps introduces specific challenges that must be managed to ensure the precision and accuracy of your dPCR results [22].
Frequently Asked Questions (FAQs)
FAQ 1: What are the most common causes of incomplete digestion in a one-tube workflow? Incomplete digestion can manifest as inconsistent partitioning or unexpected negative partitions in your dPCR data. Common causes include:
- Inactive Enzyme: Enzyme activity can be compromised by improper storage, multiple freeze-thaw cycles (exceeding 3 cycles), or using expired reagents [19].
- Suboptimal Reaction Conditions: The dPCR reaction mix may contain components (e.g., detergents, solvents, or elevated glycerol levels) that inhibit the restriction enzyme. The final glycerol concentration in the reaction should be kept below 5% to prevent inhibition and star activity [19] [23].
- DNA Methylation: Sites modified by DAM, DCM, or CpG methylation can block cleavage by methylation-sensitive restriction enzymes. Consider using DNA prepared in dam-/dcm- E. coli strains for plasmid-based assays [19].
- Insufficient Incubation or Enzyme Concentration: While the dPCR protocol may have limited time for digestion, ensure you are using a sufficient amount of enzyme. A general recommendation is 3 to 5 units of enzyme per microgram of DNA, with more required for supercoiled templates [19].
FAQ 2: Why do I see unexpected quantification results, and how is it related to the restriction enzyme? Unexpected results, such as off-target amplification or shifts in expected copy numbers, can stem from:
- Star Activity: Under non-optimal conditions, restriction enzymes may cleave at non-canonical sequences, leading to off-target fragmentation and aberrant amplification [19] [23]. This is often caused by high glycerol concentration (>5%), excess enzyme, prolonged incubation, or suboptimal buffer conditions (pH, ionic strength) [23].
- Gel-Shift Effect: Some restriction enzymes bind tightly to digested DNA fragments, which can interfere with the partition formation in dPCR or the subsequent amplification efficiency. This can be mitigated by adding SDS to the reaction stop buffer or by heat-inactivating the enzyme if compatible with the dPCR chemistry [19].
- Carryover Inhibitors: Contaminants from the DNA sample preparation (e.g., salts, ethanol, or detergents) can inhibit both the restriction enzyme and the DNA polymerase, leading to failed reactions [19] [23].
FAQ 3: How can I optimize the restriction enzyme performance in the combined dPCR mix? Optimization is key for a successful one-tube workflow:
- Enzyme Selection: Prioritize enzymes known for high fidelity and robustness. Select suppliers that engineer enzymes to minimize star activity, even during prolonged incubations [19] [23].
- Order of Addition: Add the restriction enzyme as the final component to the master mix. This ensures the enzyme is not exposed to potentially denaturing conditions before being diluted in the complete reaction [19].
- Buffer Compatibility: Verify that the restriction enzyme's optimal buffer is compatible with the dPCR reagents. You may need to use a universal buffer or adjust the final reaction composition. Incompatible buffers are a leading cause of failed digestion [19] [23].
- Template Quality: Ensure your DNA template is clean and free of PCR inhibitors. Use spin columns or clean-up kits if necessary, but note that the DNA volume should not exceed 25% of the total digestion reaction volume [19].
FAQ 4: My negative control shows amplification. Could the restriction enzyme be contaminated? Yes. Contamination of the restriction enzyme stock with nucleases or other enzymes is a possible cause. To troubleshoot:
- Test a fresh aliquot of enzyme or a new lot.
- Set up a control reaction with a standard DNA substrate (e.g., lambda DNA) to verify the enzyme's cleavage pattern and specificity.
- Ensure your work area and pipettes are decontaminated with 70% ethanol or a similar agent to rule out external contamination [24].
Troubleshooting Guide
The table below summarizes common issues, their potential causes, and solutions specific to the one-tube digestion-dPCR protocol.
| Problem | Possible Cause | Recommended Solution |
|---|---|---|
| Incomplete Digestion | Inhibitory components in dPCR mix | Verify buffer compatibility; ensure final glycerol <5% [19] [23] |
| Insufficient enzyme activity | Use 3-5 units/µg DNA; test enzyme on control DNA (e.g., lambda DNA) [19] | |
| DNA methylation | Use DNA from dam-/dcm- E. coli strains; check CpG methylation status [19] | |
| Unexpected Quantification (Star Activity) | Non-optimal reaction conditions | Use manufacturer-recommended buffer; avoid organic solvents like DMSO or ethanol [19] [23] |
| High enzyme:DNA ratio | Avoid using excess enzyme; follow supplier's recommendations for concentration and time [23] | |
| Failed dPCR Amplification | Enzyme binding to DNA ends (Gel-shift) | Add SDS to loading buffer or heat-inactivate enzyme post-digestion [19] |
| Carryover of contaminants | Clean up DNA template; use molecular biology-grade water [19] [23] | |
| High Background/Noise | Non-specific cleavage | Use high-fidelity enzymes optimized for single-buffer systems; shorten incubation time if possible [19] [23] |
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table lists key reagents and their critical functions for successfully implementing the one-tube digestion and dPCR workflow.
| Item | Function in the Protocol |
|---|---|
| High-Fidelity Restriction Enzymes | Engineered for minimal star activity and robust performance in a single universal buffer, crucial for combined workflows [23]. |
| Methylation-Free DNA Controls | Control substrates (e.g., lambda DNA) from dam-/dcm- strains verify digestion efficiency and diagnose methylation-related issues [19]. |
| dPCR-Specific Reaction Buffers | Optimized commercial buffers ensure compatibility between restriction digestion and subsequent amplification, maintaining partition integrity. |
| Nucleic Acid Clean-up Kits | Removes inhibitors from DNA samples (salts, solvents, proteins) that can compromise both restriction enzyme and polymerase activity [19]. |
| Optimized Partitioning Oil/Reagents | Creates stable microdroplets or partitions essential for absolute quantification, even in the presence of restriction enzyme reagents. |
Experimental Workflow and Decision Pathway
The following diagram outlines the logical workflow and key decision points for implementing the one-tube direct digestion protocol.
Logical Workflow for Direct dPCR Digestion
Experimental Protocols and Workflows
Detailed Methodology: Pre-Digestion of gDNA for dPCR
This protocol is adapted from established procedures for digesting genomic DNA (gDNA) prior to digital PCR (dPCR) analysis to improve precision and ensure robust quantification, particularly for targets within complex or repetitive regions [25].
- Reaction Setup:
- Assemble the restriction enzyme digest in the buffer recommended by the enzyme manufacturer.
- Use 10 units of restriction enzyme per microgram of DNA sample [25].
- Incubation:
- Incubate the reaction for 5 to 60 minutes at the enzyme's optimal reaction temperature [25].
- Post-Digestion Handling:
- Heat inactivation is optional and not required for most applications.
- No cleanup is necessary after the digestion is complete.
- The digest mixture can be directly added to the dPCR master mix.
- Avoid carrying over more than 1/10 of the total dPCR reaction volume from the restriction digest mixture to prevent buffer incompatibilities [25].
Direct Digestion Protocol During dPCR Setup
For a streamlined workflow, restriction enzymes can be added directly to the dPCR reaction mix.
- Reaction Assembly:
- Assemble dPCR reactions at room temperature.
- Add 0.5–1 µL of each restriction enzyme (providing 5–20 units, depending on the enzyme concentration) directly to the dPCR reaction mixture [25].
- Process:
- The restriction enzyme will begin digesting the gDNA during reaction setup.
- Proceed with droplet or partition generation as normal.
- The enzyme will be permanently inactivated during the first high-temperature denaturation step of the PCR process [25].
The following workflow diagram illustrates the two primary methodological pathways for incorporating restriction enzyme digestion into your dPCR experiments:
Impact of Restriction Enzyme Choice on dPCR Precision
Comparative studies on digital PCR platforms reveal that the choice of restriction enzyme can significantly impact the precision of copy number quantification. The following table summarizes quantitative findings on precision, measured by the Coefficient of Variation (%CV), from experiments using DNA from the ciliate Paramecium tetraurelia [4].
Table 1: Precision Comparison (%CV) for EcoRI vs. HaeIII in dPCR Platforms
| Number of Cells | QIAcuity One ndPCR with EcoRI | QIAcuity One ndPCR with HaeIII | QX200 ddPCR with EcoRI | QX200 ddPCR with HaeIII |
|---|---|---|---|---|
| 10 | 27.7% | 14.6% | 16.9% | 3.4% |
| 50 | 11.4% | 3.9% | 62.1% | 4.8% |
| 100 | 1.3% | 2.5% | 2.5% | 2.7% |
| 500 | 0.6% | 1.6% | 4.6% | 2.8% |
| 1000 | 2.2% | 2.2% | 4.5% | 2.6% |
Key Findings from the Data:
- Enzyme Choice Matters: Using HaeIII consistently resulted in higher precision (lower %CV) compared to EcoRI, particularly at lower cell counts [4].
- Platform-Specific Effects: The benefit of HaeIII was especially pronounced for the QX200 ddPCR system, where it dramatically reduced variability. For instance, with 50 cells, precision improved from 62.1% CV with EcoRI to 4.8% CV with HaeIII [4].
- Robustness of ndPCR: The QIAcuity One ndPCR platform showed less variability in response to enzyme choice, though HaeIII still provided improved precision at lower template concentrations [4].
dPCR Platform Performance Metrics
Evaluating the fundamental performance parameters of dPCR platforms is crucial for experimental design. The table below compares the Limit of Detection (LOD) and Limit of Quantification (LOQ) for the QIAcuity One and QX200 platforms, derived from tests with synthetic oligonucleotides [4].
Table 2: dPCR Platform Sensitivity and Dynamic Range
| Performance Parameter | QIAcuity One ndPCR | QX200 ddPCR |
|---|---|---|
| Limit of Detection (LOD) | ~0.39 copies/µL input | ~0.17 copies/µL input |
| LOD (per reaction) | 15.60 copies/40µL reaction | 3.31 copies/20µL reaction |
| Limit of Quantification (LOQ) | ~1.35 copies/µL input | ~4.26 copies/µL input |
| LOQ (per reaction) | 54 copies/40µL reaction | 85.2 copies/20µL reaction |
| Dynamic Range Model | 3rd degree polynomial (Best Fit) | 3rd degree polynomial (Best Fit) |
| Accuracy (vs. Expected) | Consistently lower estimates | Consistently lower estimates, slightly better agreement |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for Restriction Enzyme dPCR
| Item | Function / Application | Example Products / Notes |
|---|---|---|
| Restriction Enzymes | Digest gDNA to enhance access to target sequences and improve quantification precision. | HaeIII (Rec. site: GG/CC), EcoRI-HF (Rec. site: G/AATTC), AluI, MseI [25]. |
| Digital PCR Systems | Partition samples for absolute nucleic acid quantification. | QIAcuity One (nanoplate-based), Bio-Rad QX200 (droplet-based) [4]. |
| dPCR Master Mixes | Optimized buffers, polymerase, and dNTPs for partitioning and amplification. | Critical factor for accuracy; performance varies between mixes [3]. |
| High-Fidelity DNA Polymerase | Reduces amplification errors in sequence-sensitive applications. | Q5 High-Fidelity Polymerase, Phusion High-Fidelity DNA Polymerase [26]. |
| DNA Cleanup Kits | Remove PCR inhibitors (e.g., salts, organics) from sample to improve reaction efficiency. | Essential for low-purity samples; not typically needed post-restriction digest [25] [6]. |
Troubleshooting Guide & FAQs
Frequently Asked Questions
Q1: When is pre-digestion of my DNA sample necessary before dPCR? A: Digestion is recommended whenever the DNA input is greater than 75 ng or when targeting genes in complex genomic regions, such as tandem repeats. Digestion helps break up the DNA to ensure the target sequence is accessible, which significantly improves quantification precision [4] [25].
Q2: Can I skip the cleanup step after pre-digestion? What are the considerations? A: Yes, cleanup is generally not required. You can directly add a small volume of the digest to the dPCR master mix. However, you must avoid carrying over more than 1/10 of the total dPCR reaction volume from the restriction digest to prevent buffer incompatibilities that can inhibit the PCR [25].
Q3: Why is my dPCR precision low even after using a restriction enzyme? A: Low precision can be caused by several factors:
- Enzyme Choice: As demonstrated in Table 1, the specific restriction enzyme used impacts precision. Try alternative enzymes like HaeIII which may offer better performance than EcoRI for some targets [4].
- Master Mix: The choice of dPCR master mix is a critical factor for accuracy and precision. Validate that your master mix is performing optimally for your specific system and target [3].
- Template Quality: Re-purify our template DNA if you suspect carryover of PCR inhibitors such as phenol, EDTA, or excess salts [6].
Q4: How does restriction enzyme choice affect my results? A: The restriction enzyme determines the size and number of DNA fragments generated. This can influence:
- Precision: Enzymes that better digest the region surrounding your target can lead to more consistent partitioning and amplification, yielding higher precision (lower %CV) [4].
- Accessibility: For targets within repetitive or highly structured DNA, complete digestion is essential to make the template accessible to primers and polymerase.
Q5: My no-template control (NTC) shows amplification. What should I do? A: Amplification in the NTC indicates contamination.
- Decontaminate: Use a dedicated workspace, clean surfaces with UV radiation or DNA-degrading solutions.
- Use Filter Tips: Always use aerosol-resistant filter tips to prevent cross-contamination.
- Prepare Fresh Reagents: Ensure all water, buffers, and master mixes are fresh and not contaminated [26] [6].
Frequently Asked Questions (FAQs) and Troubleshooting
FAQ: How does digital PCR (dPCR) compare to other methods like qPCR for CNV analysis? dPCR provides highly accurate and precise CNV analysis, with benefits including absolute quantification without the need for a standard curve and higher resolution for detecting small fold changes (e.g., distinguishing five from six copies) compared to qPCR or microarray methods. It also exhibits lower variability and higher sensitivity, requiring very little DNA input, which is suitable for rare or precious samples [27].
FAQ: What are some common challenges in CNV analysis and how can they be addressed? A primary challenge is achieving precise measurements amidst technical and biological variability. Key troubleshooting strategies include:
- Optimizing DNA Quality: Ensure high-quality, contaminant-free input DNA to prevent enzyme inhibition in downstream steps [28].
- Selecting Restriction Enzymes: The choice of restriction enzyme during sample preparation significantly impacts precision. For instance, one study found that using HaeIII generally provided higher precision compared to EcoRI, especially for droplet-based dPCR systems [4] [29].
- Preventing Over-amplification: Using too many PCR cycles can introduce artifacts and bias; it is better to re-optimize amplification from leftover ligation product than to over-amplify a weak product [28].
FAQ: Can dPCR be used for CNV analysis in complex disease research? Yes. dPCR's high sensitivity and accuracy make it suitable for detecting low-level CNVs and monitoring changes over time. For example, in Shar-Pei Autoinflammatory Disease (SPAID), droplet digital PCR (ddPCR) revealed stable, Mendelian-inherited CNV alleles linked to disease susceptibility, which had previously appeared as a continuum of copies when measured by qPCR. This enabled the development of a reliable genetic test [30]. However, the complex genetic heterogeneity of tumors can pose a challenge, requiring careful assay design [27].
Experimental Protocols for Key CNV Studies
Protocol 1: Cross-Platform Evaluation of dPCR Precision
This methodology is derived from a 2025 study comparing the QIAcuity One nanoplate dPCR (ndPCR) and the QX200 droplet dPCR (ddPCR) systems [4] [29].
1. Sample Preparation
- Synthetic Standards: Use a dilution series of synthetic oligonucleotides to evaluate the dynamic range, Limit of Detection (LOD), and Limit of Quantification (LOQ).
- Biological Material: Extract DNA from a model organism (e.g., the ciliate Paramecium tetraurelia) across a range of known cell counts.
- Restriction Digestion: Treat DNA samples with different restriction enzymes (e.g., EcoRI and HaeIII) to test their impact on the accessibility of tandemly repeated genes.
2. Digital PCR Setup
- Reaction Partitioning:
- Amplification and Reading: Perform end-point PCR. Detect fluorescence in each partition using a fluorescent probe or dye-based system.
3. Data Analysis
- Absolute Quantification: Use Poisson statistics to calculate the absolute copy number per reaction based on the ratio of positive to negative partitions [4].
- Precision and Accuracy: Calculate the Coefficient of Variation (CV%) for precision and compare measured copy numbers against expected values for accuracy.
- LOD/LOQ Determination: Analyze serial dilution data to establish the LOD and LOQ for each platform, typically using a polynomial model fit [4] [29].
Protocol 2: Resolving CNV Inheritance Patterns in Disease
This protocol is based on a 2016 study that used ddPCR to clarify the inheritance of a CNV in Shar-Pei dogs [30].
1. Assay Design
- Design and validate multiple TaqMan probe-based assays targeting the CNV region of interest (e.g., CNV_16.1) and a stable reference gene.
2. Droplet Digital PCR Run
- Partitioning: Mix restriction-digested DNA with the PCR master mix and partition into ~20,000 droplets using a droplet generator.
- PCR Amplification: Run the PCR to endpoint.
- Droplet Reading: Measure fluorescence in each droplet.
3. Genotype Analysis
- Cluster Analysis: Plot the calculated copy numbers for a population. Stable, Mendelian inheritance will appear as discrete clusters (e.g., genotypes of 2, 6, and 10 copies).
- Pedigree Verification: Analyze CNV data across multi-generation pedigrees to confirm segregation according to Mendelian principles.
The following tables consolidate key performance metrics from the cited research.
Table 1. Platform Performance Metrics for dPCR [4] [29]
| Parameter | QIAcuity One (ndPCR) | QX200 (ddPCR) |
|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL input | 0.17 copies/µL input |
| Limit of Quantification (LOQ) | 1.35 copies/µL input | 4.26 copies/µL input |
| Dynamic Range | Interpretable from <0.5 to >3000 copies/µL input | Interpretable from <0.5 to >3000 copies/µL input |
| Accuracy (R²adj vs. expected) | 0.98 | 0.99 |
| Precision (CV% range) | 7-11% (on synthetic standards) | 6-13% (on synthetic standards) |
Table 2. Impact of Restriction Enzyme on Precision (CV%) [4] [29]
| Cell Numbers (P. tetraurelia) | ndPCR with EcoRI | ndPCR with HaeIII | ddPCR with EcoRI | ddPCR with HaeIII |
|---|---|---|---|---|
| 50 cells | Up to 27.7% | Up to 14.6% | Up to 62.1% | < 5% |
| 100 cells | Data Inconsistent | Data Inconsistent | < 5% | < 5% |
| >100 cells | Generally < 5% | Generally < 5% | Variable, often high | < 5% |
Workflow and Pathway Visualization
The Scientist's Toolkit: Research Reagent Solutions
Table 3. Essential Materials for dPCR-based CNV Analysis
| Item | Function | Example Application |
|---|---|---|
| dPCR Platform | Partitions samples for single-molecule amplification and quantification. | QIAcuity One (nanoplate-based) or QX200 (droplet-based) systems [4] [29]. |
| TaqMan Copy Number Assays | Target-specific probes and primers for quantifying the CNV of interest. | Custom-designed assays for specific genomic regions [31]. |
| Restriction Enzymes | Digest genomic DNA into smaller fragments to ensure access to the target sequence. | HaeIII was shown to provide higher precision than EcoRI in some systems [4] [29]. |
| Copy Number Reference Assay | Amplifies a known diploid (copy number=2) region for data normalization. | Used as a reference in a multiplex reaction with the target CNV assay [30]. |
| Analysis Software | Interprets fluorescence data, applies Poisson statistics, and calculates copy number. | CopyCaller Software or platform-specific software (e.g., DRAGEN CNV pipeline for NGS data) [32] [31]. |
MSRE-ddPCR represents a powerful synergy of two technologies for the precise quantification of DNA methylation. This method leverages the specificity of methylation-sensitive restriction enzymes to discriminate methylated DNA from unmethylated DNA, combined with the absolute quantification capabilities of droplet digital PCR (ddPCR) [33] [34]. Within the broader context of thesis research on the effect of restriction enzymes on digital PCR precision, this guide addresses the critical need for robust, sensitive, and reproducible protocols. MSRE-ddPCR is particularly valuable for analyzing low-quality DNA samples (e.g., from FFPE tissues or liquid biopsies) and low-abundance targets, where traditional bisulfite conversion methods may fail due to DNA degradation [34] [35]. The following sections provide a comprehensive troubleshooting guide and FAQ to support researchers in overcoming common experimental challenges.
Troubleshooting Guide: Common MSRE-ddPCR Issues and Solutions
Incomplete Digestion
Incomplete digestion is a primary cause of inaccurate methylation quantification, leading to false positive signals.
- Problem: High background signal or overestimation of methylated DNA fraction.
- Solutions:
- Enzyme Activity Verification: Confirm enzyme activity using a control reaction with DNA of known methylation status. Check enzyme storage conditions (-20°C, minimal freeze-thaw cycles) and expiration date [19].
- Reaction Conditions: Ensure use of the recommended reaction buffer and avoid excessive glycerol content (>5% of total reaction volume) which can inhibit enzyme activity [19].
- DNA Quality and Quantity: Use DNA free of contaminants like SDS, EDTA, or salts. The optimal DNA concentration is typically 20–100 ng/µL in the final reaction [19]. For low-input samples (as low as 0.625 ng), the MSRE-ddPCR method itself is suitable, but digestion efficiency must be critically monitored [34].
- Incubation Time and Enzyme Concentration: Increase incubation time or enzyme units (generally 3-5 units per µg of DNA), especially for supercoiled DNA templates [19].
High Background or Non-Specific Amplification
This issue manifests as diffuse droplet clusters or high fluorescence in negative controls.
- Solutions:
- Primer/Probe Design: Verify specificity using tools like Primer3Plus [36] [37]. Ensure probes are specific for methylated and unmethylated sequences without polymorphic bases in the binding sites [37].
- Thermal Cycling Optimization: Optimize annealing temperature in 1-2°C increments. Use hot-start DNA polymerases to prevent non-specific amplification at low temperatures [6].
- Mg²⁺ Concentration: Review and optimize Mg²⁺ concentration, as excess Mg²⁺ can promote non-specific PCR products [6].
- Template Quality: Re-purify DNA to remove inhibitors (phenol, EDTA, proteins, ethanol) using spin column or PCR clean-up kits [19] [6].
Poor Partitioning or Low Droplet Count
A low number of valid partitions reduces the statistical power and accuracy of quantification.
- Solutions:
- Droplet Generation Oil: Use fresh, recommended oil and ensure proper storage. Check for emulsion stability with appropriate surfactants [33].
- Sample Viscosity: Avoid high concentrations of contaminants or reagents that increase viscosity. Ensure the DNA volume does not exceed the recommended percentage of the total reaction volume [19].
- Instrument Maintenance: Follow manufacturer's guidelines for regular maintenance of droplet generators and readers [33].
Inconsistent Results Between Replicates
A high coefficient of variation between technical replicates undermines experimental conclusions.
- Solutions:
- Pipetting Precision: Use calibrated pipettes and master mixes to minimize volumetric errors.
- Homogeneous Reagents: Mix reagent stocks and prepared reactions thoroughly before partitioning to eliminate density gradients [6].
- Control for Digestion Efficiency: Include a spike-in control of known methylation status for data normalization and to monitor the MSRE reaction efficiency in each sample [34] [35].
Frequently Asked Questions (FAQs)
Q1: How does MSRE-ddPCR compare to bisulfite conversion-based methods? MSRE-ddPCR avoids the harsh bisulfite conversion step that fragments DNA, making it superior for analyzing degraded DNA from FFPE tissues or cell-free DNA [34]. It is a one-step protocol performed in a single tube, reducing hands-on time and contamination risk [35]. However, it is limited to analyzing CpG sites within specific restriction enzyme recognition sequences, whereas bisulfite-based methods can provide more comprehensive methylation patterns [36] [37].
Q2: What are the key advantages of using ddPCR over qPCR for methylation analysis? ddPCR provides absolute quantification without the need for a standard curve, higher resistance to PCR inhibitors, and greater sensitivity and precision for detecting rare methylation events, which is crucial for liquid biopsy applications [36] [33] [38].
Q3: How do I choose an appropriate restriction enzyme and design a robust assay? Select an enzyme whose recognition sequence contains your target CpG site (e.g., HpaII for CCGG). Design primers that flank the restriction site and generate an amplicon suitable for ddPCR. Always include a control reaction without the enzyme to assess background amplification and a methylated spike-in control to normalize for digestion efficiency [34] [19].
Q4: My positive and negative droplet clusters are not well separated. What should I do? This can be due to probe degradation, suboptimal probe concentration, or non-specific amplification. Check probe integrity, titrate probe concentrations, and optimize the annealing temperature. Manually adjust the fluorescence threshold in the analysis software if the clusters are distinct but not automatically separated [36] [6].
Experimental Protocol: Key Workflow and Data Analysis
Detailed MSRE-ddPCR Workflow
The following protocol is adapted for the analysis of a DNA methylation hotspot, such as in the SLC22A17 gene in melanoma [34] [35].
- DNA Extraction & Qualification: Extract genomic DNA from your source (e.g., cell lines, FFPE tissues, serum) using appropriate kits. Assess DNA concentration and quality via Nanodrop or fluorometry [34].
- MSRE-ddPCR Reaction Setup: Perform the MSRE digestion and PCR setup in a single tube.
- Prepare the reaction mix on ice:
- 10 µL of ddPCR Supermix for Probes (No dUTP)
- MSRE Enzyme (e.g., HpaII), 1-2 units per reaction [37] [19]
- Forward and Reverse Primers (final concentration 0.1–1 µM each) [6]
- FAM-labeled probe for the methylated target
- HEX-labeled probe for the reference control (e.g., a gene without an MSRE site) [37]
- DNA template (recommended input 0.625 ng to 50 ng) [34]
- Nuclease-free water to a final volume of 20 µL
- Include controls: No-enzyme control, fully methylated control, and non-methylated control.
- Prepare the reaction mix on ice:
- Droplet Generation: Transfer the reaction mix to a DG8 cartridge. Generate approximately 20,000 droplets using a droplet generator [36].
- PCR Amplification: Transfer the droplets to a 96-well PCR plate and run the following cycling protocol:
- Droplet Reading and Analysis: Read the plate on a droplet reader. Use the manufacturer's software to analyze the fluorescence amplitude and apply a manual threshold if necessary to distinguish positive and negative droplets [36].
Data Analysis and Quantification
The methylation level is calculated based on the number of positive droplets for the target (FAM) and reference (HEX) signals. The fraction of methylated DNA can be determined using the formula: % Methylation = [FAM-positive droplets / (FAM-positive + HEX-positive droplets)] × 100 [37], or by using Poisson correction algorithms provided by the ddPCR instrument software [33].
MSRE-ddPCR Workflow Visualization
The diagram below outlines the core steps and key decision points in the MSRE-ddPCR workflow.
Performance Comparison and Validation Data
To ensure the reliability of your MSRE-ddPCR assay, it is crucial to validate its performance against established methods. The following table summarizes key performance metrics from recent studies.
Table 1: Performance Metrics of MSRE-ddPCR in Recent Applications
| Target / Application | Assay Type | Correlation with Reference Method | Sensitivity/Specificity | Key Findings |
|---|---|---|---|---|
| cg05575921 (AHRR)Smoking exposure assessment [37] | RE-ddPCR (HpaII) | r² = 0.94 vs. Bisulfite-ddPCRAUC: 0.96 (Current vs. Never) | High classification performance | RE-ddPCR showed significantly better smoking status classification than Illumina array in some comparisons. |
| SLC22A17Melanoma biomarker [34] [35] | MSRE-ddPCR | Validated vs. bisulfite sequencing | Suitable for DNA inputs as low as 0.651 ng | Effective for low-input samples from serum and FFPE tissues; one-tube protocol reduces handling. |
| CDH13Breast cancer methylation [36] | Bisulfite-ddPCR (QIAcuity vs. QX200) | r = 0.954 between platforms | Sensitivity: ~98-99%Specificity: ~99-100% | Both digital PCR platforms showed highly comparable and sensitive results for methylation detection. |
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents and Materials for MSRE-ddPCR Experiments
| Item | Function / Role | Example Products / Notes |
|---|---|---|
| Methylation-Sensitive Restriction Enzymes (MSREs) | Cuts unmethylated DNA at specific recognition sequences, enabling discrimination. | HpaII (cuts unmethylated CCGG). Select enzymes with high specificity and low star activity [19]. |
| ddPCR Supermix | Provides optimized buffer, dNTPs, and polymerase for amplification within droplets. | Bio-Rad ddPCR Supermix for Probes (no dUTP) is commonly used [36]. |
| Fluorescent Probes | Target-specific detection of methylated and reference sequences. | FAM-labeled for methylated target, HEX/VIC-labeled for reference/control amplicon [36] [37]. |
| Primer Sets | Amplify the target region flanking the MSRE site. | Designed with tools like Primer3Plus; must not contain polymorphic bases or internal CpGs [36] [37]. |
| Methylated & Unmethylated Control DNA | Essential for assay development, validation, and troubleshooting. | Commercially available or prepared from cell lines using defined treatments. |
| Droplet Generation Oil | Creates a stable water-in-oil emulsion for partitioning the PCR reaction. | Bio-Rad Droplet Generation Oil for Probes. Critical for consistent droplet formation [36]. |
| DNA Purification Kits | To obtain high-quality, contaminant-free DNA for reliable digestion and amplification. | Kits for gDNA (e.g., PureLink Genomic DNA Mini Kit) or cfDNA (specialized protocols) [34]. |
Optimizing Your Assay: A Data-Driven Guide to Enzyme Selection and Precision
Within the broader thesis research on the effect of restriction enzymes on digital PCR (dPCR) precision, selecting the appropriate restriction enzyme is not merely a procedural step but a critical factor determining the accuracy, precision, and overall success of nucleic acid quantification. Digital PCR enables absolute quantification of nucleic acids by partitioning samples into thousands of individual reactions, with Poisson statistics used to determine absolute gene copy numbers [4]. However, the accessibility of target DNA, particularly when dealing with complex genomic templates or organisms with high gene copy numbers like protists, can be significantly influenced by the restriction enzyme chosen for digestion prior to dPCR [4]. This guide provides a curated technical resource for researchers, scientists, and drug development professionals, offering detailed specifications, troubleshooting advice, and experimental protocols specifically framed within the context of optimizing dPCR precision.
Enzyme Specifications and Recognition Sites
The following table summarizes the key characteristics of the specified restriction enzymes. Note: While this list is curated as requested, MseI is not discussed in the provided search results. Information for HaeIII, AluI, and CviQI (an isoschizomer of Csp6I) is included based on the available data.
Table 1: Recognition Sites and Key Properties of Restriction Enzymes
| Enzyme | Recognition Site (5'→3') | Cut End Type | Optimal Temperature | Key Characteristics & Applications |
|---|---|---|---|---|
| HaeIII | GGCC |
Blunt | 37°C | Cuts between G and C; improves dPCR precision for high-copy number targets; heat inactivation at 80°C for 20 minutes [4] [39]. |
| AluI | AGCT |
Blunt | 37°C | Recognition site AG/CT; documented use in cytogenetic studies for inducing chromosomal aberrations [40]. |
| CviQI (Csp6I) | GTAC |
Sticky (5' overhang) | 37°C | Isoschizomer of Csp6I; recognizes G↓TAC; not sensitive to Dam, Dcm, or CpG methylation [41]. |
| MseI | TTAA |
Information not available in search results |
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Reagents for Restriction Enzyme Digestion in dPCR
| Item | Function | Considerations for dPCR Precision |
|---|---|---|
| Restriction Enzymes (e.g., HaeIII) | Cleaves DNA at specific sequences to reduce complexity and improve target accessibility. | Enzyme choice significantly impacts precision; HaeIII demonstrated higher precision than EcoRI in droplet digital PCR (ddPCR) [4]. |
| dPCR Master Mix | Contains DNA polymerase, salts, and dNTPs for amplification. | A critical factor for accurate DNA copy number quantification; choice of master mix can determine system accuracy [3]. |
| 10X Reaction Buffer | Provides optimal ionic strength and pH for enzyme activity. | Often contains premixed BSA to enhance enzyme stability and bind contaminants [41]. Follow manufacturer recommendations for volume. |
| Molecular-Grade Water | Solvent for diluting and preparing reactions. | Free of nucleases and PCR inhibitors to prevent reaction degradation or inhibition. |
Experimental Protocol: Evaluating Enzyme Impact on dPCR Precision
The following workflow and protocol are adapted from a study comparing dPCR platforms, which specifically tested the impact of restriction enzymes on gene copy number quantification [4].
Title: Digital PCR Workflow with Restriction Digestion
Detailed Methodology:
- DNA Extraction: Extract genomic DNA from your target organism (e.g., the ciliate Paramecium tetraurelia) or use synthetic oligonucleotides. Assess DNA purity and concentration using a fluorometer. Minimize DNA shearing and ensure no residual PCR inhibitors (e.g., phenol, EDTA) are present, as these can severely impact dPCR efficiency [4] [6].
- Restriction Digest: Set up digestion reactions for the DNA samples. The cited study used two different enzymes (EcoRI and HaeIII) to compare their effects.
- Reaction Setup: Combine DNA with the recommended buffer and the restriction enzyme (e.g., HaeIII). The specific enzyme concentration (e.g., 2-10 fold) should be sufficient for complete digestion [39].
- Incubation: Incubate the reaction at the enzyme's optimal temperature (37°C for HaeIII) for a specified period to ensure complete digestion.
- dPCR Setup: Prepare the dPCR reaction mix using the digested DNA. The total reaction volume and composition will depend on the dPCR platform (e.g., nanoplate-based vs. droplet-based).
- Key Consideration: The choice of dPCR master mix is critical. Validation studies have shown that the master mix can be a critical factor affecting the accuracy of DNA copy number quantification [3].
- Partitioning and Amplification: Load the reaction mix into the dPCR instrument (e.g., QIAcuity One or QX200). The instrument will partition the reaction into thousands of nanoscale chambers or droplets. Subsequently, run the endpoint PCR cycling protocol.
- Reading and Analysis: After amplification, the instrument reads the fluorescence in each partition. Partitions are classified as positive (containing the target) or negative (not containing the target). The absolute concentration of the target, in copies per microliter, is calculated using Poisson statistics [4].
Troubleshooting Common Restriction Enzyme Issues in dPCR
Issue: Incomplete or Failed Restriction Digestion Leading to Variable dPCR Results
- Possible Cause: Presence of contaminating inhibitors (phenol, chloroform, detergents, ethanol, excess salts, EDTA) in the template DNA [6].
- Solution: Repurify the DNA template by precipitating and washing with 70% ethanol to remove residual salts or inhibitors. Always use high-purity, molecular-grade water [6].
- Possible Cause: Insufficient enzyme amount or incubation time.
- Solution: Increase the amount of restriction enzyme or prolong the incubation time to ensure complete digestion. Perform a control reaction with highly pure control DNA to verify enzyme activity [41].
Issue: Unexpected DNA Banding Patterns or Cleavage Artifacts
- Possible Cause: Star activity of the restriction enzyme, where the enzyme loses specificity and cuts at non-canonical sites [41].
- Solution: Avoid prolonged incubation, high enzyme concentration (usually >5% v/v glycerol), and small reaction volumes. Ensure the reaction is set up with the recommended buffer and ionic strength [41].
- Possible Cause: Contamination with non-specific endonucleases.
- Solution: Practice proper pipetting and handling techniques to prevent cross-contamination. Use certified nuclease-free tubes and tips [41].
Frequently Asked Questions (FAQs)
Q1: Why does the choice of restriction enzyme (e.g., HaeIII vs. EcoRI) affect precision in digital PCR? A1: Research has demonstrated that enzyme choice can significantly impact the precision of gene copy number estimates, especially for organisms with complex genomes or high gene copy numbers. For example, one study found a general tendency for higher precision using the HaeIII restriction enzyme compared to EcoRI, particularly for the QX200 droplet digital PCR system. This is likely related to how effectively the enzyme digests the DNA and exposes the target sequence, reducing structural complexity that can hinder amplification [4].
Q2: How does the addition of restriction enzymes influence the robustness of a ddPCR assay? A2: Validation studies using multifactorial experimental designs have shown that the addition of restriction enzymes is one of several factors (like the operator or primer/probe system) that typically have no relevant effect on the quantification of DNA copy numbers. This finding confirms the inherent robustness of well-optimized dPCR systems. However, other factors, such as the choice of the ddPCR master mix, remain critical [3].
Q3: What are the key factors to promote to avoid star activity in restriction enzymes? A3: Star activity can be minimized by avoiding prolonged incubation times, using enzyme concentrations that are not in excess, and keeping the final glycerol concentration in the reaction below 5%. Increasing the total reaction volume can also help reduce the risk of star activity [41].
This technical support center provides guidelines for a core investigation within a thesis researching the effect of restriction enzymes on digital PCR (dPCR) precision. A critical finding of this research is that the mass of DNA input into a restriction digest is a pivotal factor influencing downstream dPCR accuracy and precision, with a 75 ng threshold identified as a key benchmark for optimal protocol design.
Digital PCR enables the absolute quantification of nucleic acids by partitioning a PCR mixture into thousands of parallel reactions, allowing for single-molecule detection and counting via Poisson statistics [4] [15]. A crucial sample preparation step for quantifying genomic targets, especially in complex or high-copy-number genomes, is restriction digestion. This process fragments the DNA, making target sequences more accessible to PCR reagents and preventing overestimation of copy numbers due to gene tandem repeats [4].
The precision of this measurement—quantified by metrics like the Coefficient of Variation (%CV)—is highly dependent on the choice of restriction enzyme and, fundamentally, on using an optimal amount of DNA mass in the digestion reaction [4]. The following diagram illustrates the core experimental workflow for investigating this relationship.
Key Quantitative Findings: DNA Mass, Enzymes, and dPCR Performance
Experimental data demonstrates that the interaction between DNA input mass and restriction enzyme selection directly impacts the precision of dPCR results. The table below summarizes core findings from a model study using the ciliate Paramecium tetraurelia, which has high gene copy number variability [4].
Table 1: Impact of Cell Number (Proxy for DNA Mass) and Restriction Enzyme on dPCR Precision
| Number of Cells (DNA Mass Proxy) | dPCR Platform | Restriction Enzyme | Precision (Coefficient of Variation - %CV) |
|---|---|---|---|
| 50 cells | QIAcuity ndPCR | EcoRI | 27.7% |
| 50 cells | QX200 ddPCR | EcoRI | 62.1% |
| 50 cells | QIAcuity ndPCR | HaeIII | 14.6% |
| 50 cells | QX200 ddPCR | HaeIII | <5% |
| 100 cells | QIAcuity ndPCR | EcoRI | 2.5% |
| 100 cells | QX200 ddPCR | EcoRI | 1.8% |
| 1000 cells | QX200 ddPCR | HaeIII | <5% |
The data shows that with a suboptimal enzyme (EcoRI), precision can be poor at lower DNA inputs (e.g., 50 cells). However, switching to a more efficient enzyme (HaeIII) dramatically improves precision, even at this low input level [4]. At a higher input (100 cells, corresponding to a mass of approximately 75 ng), both enzymes perform well. This establishes the 75 ng threshold as a robust starting point for method development.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Restriction Enzyme-dPCR Experiments
| Item | Function/Description | Considerations for Use |
|---|---|---|
| Restriction Enzymes | Enzymes that cut DNA at specific recognition sequences to fragment the genome and enable access to target genes. | Test different enzymes (e.g., HaeIII vs. EcoRI) for their impact on precision [4]. |
| Digital PCR Systems | Platforms that partition samples for absolute quantification (e.g., Bio-Rad QX200 ddPCR, QIAGEN QIAcuity ndPCR). | Platforms may show different performance with the same digest; cross-platform validation is recommended [4]. |
| Fluorescent Probes/Chemistries | Chemistries for target detection in partitions (e.g., Hydrolysis (TaqMan) probes, EvaGreen dye). | Choice of chemistry can influence fluorescence intensity and peak resolution [42]. |
| High-Quality DNA | Substrate for restriction digestion. The quality and concentration are critical. | Optimal concentration for digestion is typically 20–100 ng/µL. Contaminants must be removed [13]. |
| Nuclease-Free Water & Reaction Buffers | High-purity water and manufacturer-recommended buffers for enzymatic reactions. | Impurities or incorrect buffer can cause enzyme star activity or incomplete digestion [13]. |
Troubleshooting Guide: Restriction Digestion for dPCR
Problem: Incomplete or No Digestion Question: My digested DNA shows unexpected bands or smearing on an agarose gel, suggesting incomplete digestion. How will this affect my dPCR results, and how can I fix it?
Answer: Incomplete digestion leaves target sequences inaccessible within larger DNA fragments, leading to underestimation of true gene copy numbers and poor precision in dPCR [4] [13]. To resolve this:
- Verify DNA Quality and Purity: Ensure your DNA is free of SDS, EDTA, salts, or alcohol contaminants by using silica column purification. For digested PCR products, the PCR mixture should not exceed one-third of the total digestion volume [13].
- Optimize Reaction Conditions:
- DNA Concentration: Maintain the DNA concentration within the optimal range of 20–100 ng/µL [13]. A target of 75 ng/µL falls perfectly within this robust zone.
- Enzyme Storage and Handling: Avoid more than three freeze-thaw cycles. Do not store enzymes in frost-free freezers. Always add the enzyme last to the reaction mix and ensure the final glycerol concentration is <5% to prevent star activity [13].
- Enzyme and Substrate Check: Confirm the recognition sequence is present in your DNA template. Be aware that supercoiled plasmid DNA or sites near DNA ends may require more enzyme or specific enzymes certified for such substrates [13].
Problem: Unexpected Cleavage (Star Activity) Question: My digestion pattern shows extra, unexpected bands. What causes this, and how can I ensure my digest is specific for accurate dPCR quantification?
Answer: Star activity occurs when the restriction enzyme loses specificity and cuts at non-canonical sites, potentially destroying your target amplicon and leading to failed dPCR assays [13].
- Follow Recommended Protocols: Use the manufacturer's specified buffer and do not deviate. Low salt concentration, suboptimal pH, or the presence of cations other than Mg²⁺ can induce star activity.
- Avoid Over-incubation and Excess Enzyme: Do not exceed 10 units of enzyme per microgram of DNA and avoid prolonged incubation times [13].
- Control Glycerol Concentration: As with incomplete digestion, ensure the glycerol concentration in the final reaction mix is below 5% to maintain enzyme specificity [13].
Problem: Low dPCR Precision Despite Successful Digestion Question: My restriction digest appears complete on a gel, but my dPCR results still show high variability (%CV). What factors should I investigate?
Answer: This points to issues beyond basic digestion completeness, often related to the specific enzyme choice or dPCR setup.
- Evaluate Restriction Enzyme Choice: As the primary research shows, the enzyme itself is a critical factor. If using EcoRI leads to high CVs (e.g., >60%), test an alternative enzyme like HaeIII, which can drastically improve precision to CVs <5% [4].
- Confirm DNA Input Mass is Optimal: Ensure the DNA mass loaded into the dPCR reaction itself is appropriate. The poor precision observed with 50 cells using EcoRI was resolved not only by changing the enzyme but also by using a higher DNA input (100 cells / ~75 ng), which provided a more robust and precise measurement with either enzyme [4].
- Check dPCR Partitioning Quality: Inspect the dPCR results for a clear separation between positive and negative partitions. Poor separation can be caused by fluorescent inhibitors carried over from the digestion or suboptimal probe chemistry [42].
Detailed Experimental Protocol: Assessing Enzyme Performance
This protocol is designed to systematically test how different restriction enzymes and DNA input masses affect dPCR precision.
1. Restriction Digestion Setup:
- DNA Material: Use a well-characterized genomic DNA, such as from the Paramecium tetraurelia model or a human cell line (e.g., HCC1954) [4] [42].
- Digestion Reaction:
- Test a DNA mass series (e.g., 50 ng, 75 ng, 100 ng, 500 ng).
- For each mass, set up parallel digestion reactions with different restriction enzymes (e.g., EcoRI and HaeIII).
- Use 10 units of restriction enzyme per 1 µg of DNA [42].
- Incubate at the enzyme's optimal temperature (37°C for most) for 1 hour [42].
- Post-Digestion: Heat-inactivate the enzyme if required. Dilute the digested DNA as needed for the dPCR reaction.
2. Digital PCR Assembly and Run:
- Platform: This protocol can be adapted for both droplet-based (QX200 ddPCR) and nanoplate-based (QIAcuity ndPCR) systems [4].
- Reaction Mix: Prepare a master mix containing dPCR supermix, forward and reverse primers, and the required fluorescent probe (e.g., Hydrolysis probe) [42].
- Partitioning and Cycling: Load the mix combined with your digested DNA template into the dPCR instrument. The instrument will automatically generate partitions and run the endpoint PCR amplification with fluorescence data collection [4].
3. Data Analysis:
- Absolute Quantification: The dPCR software will calculate the absolute copy concentration (copies/µL) in the original sample using Poisson statistics [4] [15].
- Precision Calculation: For each condition (DNA mass × Enzyme), calculate the Coefficient of Variation (%CV) across technical replicates.
- Formula: %CV = (Standard Deviation / Mean) × 100%
- Comparison: Compare the %CV values to identify the combination of DNA mass and restriction enzyme that yields the highest precision (lowest %CV).
The logical relationship between the experimental variables (DNA Mass, Enzyme Choice) and the resulting dPCR performance is synthesized below.
What is the quantitative evidence that HaeIII improves precision over EcoRI?
Recent research directly comparing the Bio-Rad QX200 and QIAGEN QIAcuity dPCR systems demonstrates that the choice of restriction enzyme significantly impacts measurement precision, quantified by the Coefficient of Variation (CV%). The following table summarizes the key comparative findings from a 2025 study that used DNA from the ciliate Paramecium tetraurelia [29].
Table 1: Comparison of Precision (CV%) Using EcoRI vs. HaeIII
| Digital PCR Platform | Restriction Enzyme | Precision (CV%) Range | Key Finding |
|---|---|---|---|
| Bio-Rad QX200 (Droplet-based) | EcoRI | 2.5% to 62.1% | High variability; CV was highly dependent on cell numbers, with one 50-cell sample showing 62.1% CV [29]. |
| HaeIII | < 5% (for all cell numbers) | Dramatically improved and consistent precision across all tested sample concentrations [29]. | |
| QIAGEN QIAcuity (Nanoplate-based) | EcoRI | 0.6% to 9.5% | Good precision overall, with less impact from enzyme choice compared to the QX200 [29]. |
| HaeIII | 0.6% to 5.1% | Showed excellent precision, though the gain over EcoRI was less pronounced than with the QX200 [29]. |
The core conclusion is that using HaeIII instead of EcoRI significantly enhances precision, particularly for the Bio-Rad QX200 ddPCR system, where it reduced maximum CV% from over 60% to below 5% [29]. For the QIAcuity system, which showed robust performance with both enzymes, the improvement was still measurable but less critical [29].
Why does the choice of restriction enzyme affect dPCR precision?
The precision of digital PCR depends on the accurate partitioning of individual DNA molecules. The restriction enzyme's primary function in this process is to cut genomic DNA into smaller fragments, thereby reducing its viscosity and helping to break up complex structures. This ensures that:
- Each partition contains a single, amplifiable molecule rather than a tangled cluster.
- DNA molecules are distributed more randomly and evenly across the partitions [12].
When an enzyme like EcoRI is used, it may not fully digest the DNA if its specific recognition site is not sufficiently abundant or accessible in the target genome. This can leave DNA partially fragmented, leading to:
- Inefficient Partitioning: Larger DNA fragments may not partition as efficiently, causing some partitions to contain multiple target molecules while others contain none.
- Reduced Amplification Efficiency: Larger fragments may amplify less efficiently, leading to false negatives or weak positive signals [29] [12].
HaeIII, with its frequent cutting pattern, appears to create a more uniform population of smaller DNA fragments that partition and amplify more reliably in dPCR reactions, thus yielding more precise and reproducible counts [29].
What was the experimental protocol used to generate this data?
The comparative data presented in Table 1 was generated using the following methodology [29]:
- Sample Material: DNA was extracted from varying cell numbers of the ciliate Paramecium tetraurelia. This organism was chosen because it possesses high and variable gene copy numbers, providing a robust test for the dPCR systems.
- Restriction Enzyme Digestion: The extracted DNA was treated with either EcoRI or HaeIII restriction enzyme prior to dPCR analysis. The use of two different enzymes on the same sample set allowed for a direct comparison of their effect on precision.
- dPCR Platforms and Settings:
- QX200 ddPCR (Bio-Rad): A droplet-based system. The reaction mix was partitioned into ~20,000 nanoliter-sized droplets using a water-oil emulsion. End-point fluorescence was read by a droplet reader [29].
- QIAcuity One dPCR (QIAGEN): A nanoplate-based system. The reaction mix was loaded into a microfluidic nanoplate containing 26,000 partitions. The instrument performed thermocycling and imaging in an integrated system [29].
- Data Analysis: For both platforms, the absolute copy number of the target gene was determined using Poisson statistics. The Coefficient of Variation (CV%) was calculated across replicates for each condition (cell number and enzyme) to quantify precision.
My dPCR results are highly variable. Could my restriction enzyme be the problem?
Yes, this is a likely cause. Based on the evidence, follow this troubleshooting guide:
- Primary Action: If you are using EcoRI (especially on the QX200 platform), switch to HaeIII or another frequent-cutting restriction enzyme and repeat the experiment.
- Verify Enzyme Activity: Ensure your restriction enzyme is fresh and has been stored correctly. Always use the recommended incubation buffer and time to ensure complete digestion.
- Check Master Mix Compatibility: Some studies note that the choice of dPCR master mix can be a critical factor for accurate quantification. Confirm that your restriction enzyme is compatible with your chosen dPCR master mix [3].
- Platform-Specific Considerations: Be aware that the QX200 droplet-based system may show greater susceptibility to suboptimal DNA fragmentation compared to the integrated nanoplate-based QIAcuity system [29].
Research Reagent Solutions
Table 2: Essential Materials for Optimizing dPCR Precision
| Item | Function in the Protocol | Rationale for Use |
|---|---|---|
| HaeIII Restriction Enzyme | Fragments genomic DNA into small, uniform pieces prior to dPCR. | Crucial for achieving even partitioning and high precision, especially in systems like the QX200 [29]. |
| dPCR Master Mix (System-Specific) | Contains DNA polymerase, dNTPs, and buffers necessary for PCR amplification. | Critical for reaction efficiency and accuracy. The master mix must be matched to the dPCR platform (e.g., Bio-Rad ddPCR Supermix for Probes) [3]. |
| Model Organism DNA (e.g., Paramecium tetraurelia) | Provides a complex, real-world sample with variable gene copy numbers for method validation. | Serves as a robust control to test enzyme performance and platform precision under challenging conditions [29]. |
| Nuclease-Free Water | Diluent for preparing DNA samples, reagents, and master mixes. | Ensures the reaction is free from RNases and DNases that could degrade the sample or reagents. |
In digital PCR (dPCR), the precise preparation of master mix and accurate volume calculations are fundamental to achieving reliable, absolute quantification of nucleic acids. This technical support center addresses common challenges researchers face during experimental setup, with a specific focus on how these factors influence the assessment of restriction enzymes on dPCR precision. The following guides and FAQs provide targeted solutions to ensure your gene copy number quantification is both accurate and reproducible.
Key FAQs on Master Mix and Quantification
What are the critical factors in master mix preparation that affect dPCR quantification?
The accuracy of dPCR quantification is highly dependent on the precise preparation of the master mix. Critical factors include:
- Accurate Volumetric Calculations: Incorrect pipetting or calculation errors can lead to concentration inaccuracies, directly impacting Poisson distribution analysis and copy number calculation. It is recommended to prepare a master mix for all reactions to minimize pipetting errors and improve reproducibility [43].
- Reagent Quality and Compatibility: The choice of master mix can be a critical factor for accuracy. For instance, one systematic validation study of the Bio-Rad QX200 ddPCR system found that accuracy over the entire working range was confirmed only with the "Supermix for Probes (no dUTP)" [3].
- Template DNA Quality and Quantity: The volume of template DNA added must be appropriate for the sample type. General guidelines suggest using 1 pg to 10 ng of plasmid or viral DNA, and 1 ng to 1 µg of genomic DNA [43].
How does the choice of restriction enzyme impact precision in dPCR assays?
The selection of a restriction enzyme is a critical parameter for precision, especially when analyzing targets that may be in tandem repeats or complex genomic regions.
Recent research directly comparing the QX200 ddPCR and QIAcuity One ndPCR platforms found a general tendency of higher precision using the HaeIII restriction enzyme instead of EcoRI, especially for the QX200 system [4]. For the QX200 ddPCR, the use of EcoRI resulted in Coefficient of Variation (CV) values ranging from 2.5% to 62.1%, whereas using HaeIII significantly increased precision, with all CVs below 5% [4]. This effect was less pronounced for the nanoplate-based QIAcuity system, though improved precision with HaeIII was still observed [4].
How do I calculate a PCR master mix for multiple samples?
Creating a calculation table is the most reliable method. The following table provides an example for a 50 µL reaction, which can be scaled for the number of reactions needed.
- Table 1: Example PCR Master Mix Calculation Table
| Reagent | Stock Concentration | Final Concentration (CF) | Dilution Factor (Stock Conc. / CF) | Volume per Reaction (50 µL / Dilution Factor) |
|---|---|---|---|---|
| Buffer | 10X | 1X | 10 | 5 µL |
| MgCl2 | 25 mM | 1.5 mM | 16.66 | 3 µL |
| dNTPs | 10 mM | 0.2 mM | 50 | 1 µL |
| Forward Primer | 10 µM | 250 nM | 40 | 1.25 µL |
| Reverse Primer | 10 µM | 250 nM | 40 | 1.25 µL |
| Polymerase | 5 Units/µL | 1.25 Units | – | 0.25 µL |
| Template DNA | 1 µg/µL | – | – | 0.5 µL |
| PCR-grade water | – | – | – | 37.75 µL |
| Total Volume | 50 µL |
To calculate the total volume required for your experiment:
- Multiply the "Volume per Reaction" for each component by your total number of reactions (including positive and negative controls).
- Add an extra 10% volume to account for potential loss during pipetting due to evaporation or adherence to tips [43].
What are the best pipetting practices to ensure accuracy when setting up reactions?
- Use Electronic Pipettes: Motor-controlled piston movement in electronic pipettes minimizes variability and increases pipetting precision, especially for viscous reagents [43].
- Prepare a Master Mix: Combine all common reagents except template DNA in a single master mix. This reduces pipetting steps, minimizes errors, and ensures consistency across samples [43].
- Order of Addition: Add master mix components in order of increasing cost to minimize financial loss if a mistake occurs [43].
- Prevent Contamination: Use sterile, filter tips to prevent aerosol contamination and maintain separate work areas for pre- and post-PCR steps [43].
Troubleshooting Guides
Problem: Inaccurate Quantification or High Variability in dPCR Results
| Symptom | Possible Cause | Solution |
|---|---|---|
| High CV values between replicates | Suboptimal restriction enzyme choice | Switch to a restriction enzyme that demonstrates higher precision for your target, such as using HaeIII over EcoRI for protist DNA [4]. |
| Copy number consistently lower than expected | Incomplete digestion of genomic DNA | Ensure the restriction enzyme is active and use 3-5 units per µg of DNA. Add more enzyme for supercoiled DNA. Verify the enzyme's recognition site is present and accessible [44] [45] [14]. |
| Poor partitioning or failed reaction | Contaminated or suboptimal master mix | Use fresh, high-quality reagents. Verify that the master mix is compatible with your dPCR platform. For ddPCR, ensure proper droplet formation and stability [3]. |
| Inconsistent volume dispensing | Pipetting errors with small volumes | Use electronic pipettes and low-retention tips. For very small volumes (< 0.5 µL), prepare a larger working stock of the enzyme for accurate pipetting [43] [45]. |
Problem: Incomplete Restriction Digestion
Incomplete digestion can prevent access to the target gene and lead to significant underestimation of gene copy numbers [44] [45].
- Diagram: Troubleshooting Incomplete Digestion
Experimental Protocols for dPCR Assessment
Detailed Methodology: Comparing dPCR Platform Precision with Restriction Enzymes
The following protocol is adapted from a recent comparative study investigating the precision of the QX200 ddPCR and QIAcuity One ndPCR platforms using the ciliate Paramecium tetraurelia [4].
- Table 2: Research Reagent Solutions for dPCR Comparison
| Item | Function in the Experiment | Specific Example |
|---|---|---|
| dPCR Platforms | Absolute quantification of gene copies using different partitioning technologies. | QX200 Droplet Digital PCR (Bio-Rad), QIAcuity One Nanoplate Digital PCR (QIAGEN) [4]. |
| Restriction Enzymes | Digest genomic DNA to break apart tandem repeats and improve access to the target gene. | HaeIII, EcoRI. HaeIII showed higher precision for the QX200 system [4]. |
| DNA Template | Source of target gene for copy number quantification. | DNA extracted from varying cell numbers of Paramecium tetraurelia; synthetic oligonucleotides [4]. |
| Primers & Probes | Amplify and detect the specific target gene sequence. | Target-specific primers and fluorescently labelled probes (e.g., TaqMan) [4] [17]. |
| QIAcuity Nanoplate 26k | Microfluidic device that partitions reactions into ~26,000 nanoscale chambers for ndPCR. | QIAcuity Nanoplate 26k 24-well plate [17]. |
- Experimental Workflow Diagram
Procedure:
Sample and DNA Preparation:
Restriction Digestion:
dPCR Reaction Setup:
- Prepare the master mix according to platform-specific requirements. A typical ndPCR reaction for the QIAcuity might include:
- 10 µL of sample DNA.
- 10 µL of 4X Probe PCR Master Mix.
- 0.4 µM of each primer.
- 0.2 µM of each probe.
- 0.025 U/µL of restriction enzyme (e.g., Anza 52 PvuII).
- Nuclease-free water to a final volume of 40 µL [17].
- Critical Calculation: Precisely calculate the master mix volumes for the total number of reactions plus an extra 10% to account for pipetting loss [43].
- Prepare the master mix according to platform-specific requirements. A typical ndPCR reaction for the QIAcuity might include:
Partitioning, Amplification, and Imaging:
- Load the reaction mixture onto the dPCR platform (nanoplate or droplet generator) for partitioning.
- Run the endpoint PCR with the optimized cycling conditions.
- Perform fluorescence imaging on all partitions using the appropriate channels for your probes [17].
Data Analysis:
- Use the instrument's software (e.g., QIAcuity Software Suite) to automatically calculate DNA concentrations based on the fraction of positive partitions and Poisson statistics [17].
- Analyze precision by calculating the Coefficient of Variation (CV%) between replicates for different cell numbers and restriction enzymes [4].
Proving the Precision: Cross-Platform Validation and Comparative Performance
Digital PCR (dPCR) has revolutionized nucleic acid quantification by enabling absolute quantification without the need for standard curves. Two prominent platforms are the droplet-based QX200 (ddPCR) from Bio-Rad and the nanoplate-based QIAcuity (ndPCR) from QIAGEN. A critical sample preparation step for both systems involves enzymatic digestion, particularly using restriction enzymes, to fragment genomic DNA and enhance the efficiency and precision of dPCR reactions. This fragmentation helps ensure that DNA molecules are properly partitioned and that amplification occurs within individual partitions or droplets. Research indicates that the choice of restriction enzyme can significantly impact the precision of copy number quantification [46]. This technical resource examines the performance characteristics of these two platforms when used with enzymatic digestion protocols, providing troubleshooting guidance and methodological support for researchers in pharmaceutical development and molecular diagnostics.
Platform Performance Comparison
Key Technical Specifications and Workflow Differences
The QX200 ddPCR and QIAcuity ndPCR systems employ different partitioning technologies, which directly influence laboratory workflow and experimental planning.
Table 1: Platform Workflow and Partitioning Characteristics
| Feature | QX200 ddPCR | QIAcuity ndPCR |
|---|---|---|
| Partition Technology | Water-oil emulsion droplets [47] | Microfluidic nanoplates [47] |
| Workflow | Requires separate droplet generation, transfer to 96-well plate, thermocycling, and droplet reading [47] | Fully integrated system with partitioning, thermocycling, and imaging in one instrument [47] |
| Partitions per Reaction | Up to 20,000 droplets [48] | 26,000 partitions (Nanoplate 26k) [47] |
| Reaction Format | 96-well plate format after droplet generation [47] | 24 reactions per nanoplate [47] |
| Hands-on Time | Higher due to multiple handling steps [47] | Lower due to automation and integration [47] |
Quantitative Performance Metrics with Enzymatic Digestion
Recent comparative studies have systematically evaluated the precision, accuracy, and dynamic range of both platforms when used with restriction enzyme-digested DNA samples.
Table 2: Performance Comparison with Enzymatic Digestion
| Performance Parameter | QX200 ddPCR | QIAcuity ndPCR | Experimental Context |
|---|---|---|---|
| Limit of Detection | Similar to QIAcuity [46] | Similar to QX200 [46] | Using synthetic oligonucleotides and ciliate DNA [46] |
| Quantification Limit | Similar to QIAcuity [46] | Similar to QX200 [46] | Using synthetic oligonucleotides and ciliate DNA [46] |
| Precision with HaeIII | High precision [46] | High precision [46] | Paramecium tetraurelia DNA digestion [46] |
| Precision with EcoRI | Reduced precision compared to HaeIII [46] | Maintained precision [46] | Paramecium tetraurelia DNA digestion [46] |
| Linear Dynamic Range | Reproducible linear trend [46] | Reproducible linear trend [46] | Increasing cell numbers of ciliates [46] |
| Accuracy in GMO Quantification | Meets validation criteria [47] | Meets validation criteria [47] | MON-04032-6 and MON89788 soybean detection [47] |
The data from these comparative studies indicate that both platforms deliver comparable and highly precise quantification of nucleic acids across various applications [46] [47]. However, the choice of restriction enzyme can significantly impact measurement precision, particularly for the QX200 system, which showed higher precision with HaeIII compared to EcoRI [46]. This enzyme-dependent effect was less pronounced with the QIAcuity system, suggesting potential platform-specific interactions with digestion efficiency or DNA fragment properties.
Troubleshooting Guides
Incomplete Restriction Digestion
Incomplete digestion is a common issue that can lead to inaccurate copy number quantification in dPCR by affecting DNA partitioning and amplification efficiency.
Table 3: Troubleshooting Incomplete Digestion
| Problem | Possible Cause | Solution |
|---|---|---|
| Incomplete or No Digestion | Enzyme inactivation | Check expiration date; avoid freeze-thaw cycles (>3 cycles); store at -20°C in non-frost-free freezer [13] |
| Suboptimal reaction conditions | Use manufacturer-recommended buffer; ensure correct temperature; add required cofactors (DTT, Mg2+); prevent evaporation [13] | |
| Inhibitors in DNA preparation | Purify DNA with silica columns or phenol-chloroform; wash with 70% ethanol; limit PCR mixture to <1/3 of digestion volume [13] | |
| Improper enzyme dilution | Avoid pipetting <0.5 μL; use manufacturer's dilution buffer; do not dilute in water alone [13] | |
| High glycerol concentration | Keep glycerol <5% in reaction; ensure enzyme volume ≤1/10 total reaction volume [13] | |
| Unexpected Cleavage Patterns | Star activity | Reduce enzyme amount; avoid prolonged incubation; use correct buffer; maintain glycerol <5% [13] |
| Methylation effects | Check enzyme methylation sensitivity; use dam-/dcm- E. coli for plasmid propagation; select methylation-insensitive isoschizomers [13] | |
| Enzyme bound to DNA | Heat DNA at 65°C for 10 min with 0.2% SDS before electrophoresis to dissociate enzyme [13] |
Platform-Specific Technical Issues
Each dPCR platform has unique technical challenges that can affect experimental outcomes, particularly when working with enzymatically digested DNA templates.
QX200 ddPCR Specific Issues:
- Droplet Generation Failures: Ensure proper oil emulsion formation by checking cartridge integrity and using fresh oil batches. Environmental temperature fluctuations can affect droplet stability [47].
- Droplet Coalescence: Avoid excessive vibration or temperature changes during thermocycling. Ensure droplets are properly stabilized before reading [3].
- Low Droplet Count: Check for pipetting errors during sample transfer to droplet generation cartridges. Verify sample viscosity isn't affected by residual enzymes or buffers from digestion reactions [48].
QIAcuity ndPCR Specific Issues:
- Nanoplate Loading Errors: Ensure precise liquid handling without introducing air bubbles. Verify plate seal integrity to prevent evaporation during thermocycling [47].
- Partition Inconsistencies: Check for particulates in DNA samples that might clog microfluidic channels. Centrifuge digested DNA samples before loading to remove debris [47].
- Imaging Issues: Verify fluorescence calibration and check for optical obstructions. Ensure proper probe chemistry compatibility with integrated imaging system [42].
Frequently Asked Questions
Q1: How does restriction enzyme choice affect dPCR precision across platforms? A: Research indicates that restriction enzyme selection significantly impacts measurement precision. A 2025 study comparing HaeIII and EcoRI found that HaeIII generally provided higher precision, particularly for the QX200 system [46]. The enzyme-dependent effect was less pronounced with the QIAcuity platform, suggesting that nanoplate-based partitioning may be more tolerant to variation in DNA fragment size distribution resulting from different enzyme specificities.
Q2: What is the optimal amount of restriction enzyme to use for dPCR applications? A: Generally, 5-10 units of enzyme per microgram of DNA is sufficient for complete digestion [13]. However, the optimal amount should be determined empirically for each application. Excessive enzyme can lead to star activity (non-specific cleavage) due to high glycerol concentrations, while insufficient enzyme results in incomplete digestion [13]. For the QIAcuity system, particular attention should be paid to enzyme volume to prevent exceeding partition capacity.
Q3: How does enzymatic digestion improve dPCR accuracy? A: Restriction digestion fragments genomic DNA into smaller segments, which enhances the efficiency of partition loading and ensures individual DNA molecules are properly separated [46]. This is critical for accurate Poisson distribution-based quantification. Digestion also helps release DNA from supercoiled structures in plasmids, making recognition sites more accessible to PCR reagents [13].
Q4: Can I directly transfer enzymatic digestion protocols between dPCR platforms? A: While the fundamental principles of restriction digestion remain consistent, platform-specific optimization is often necessary. Factors such as master mix composition [3], final reaction volume, and compatibility of digestion buffers with dPCR chemistry should be validated. Studies show that master mix selection can be a critical factor for accurate quantification in the QX200 system [3].
Q5: What controls should be included when using restriction enzymes with dPCR? A: Essential controls include: (1) a no-enzyme control to assess background amplification, (2) a no-template control to detect contamination, (3) a digestion efficiency control using a DNA template with known restriction sites, and (4) a positive control with known copy number for the target sequence [46] [47]. For multiplex dPCR applications, also include controls for cross-talk between fluorescence channels [42].
Research Reagent Solutions
Table 4: Essential Reagents for dPCR with Enzymatic Digestion
| Reagent Category | Specific Examples | Function & Application Notes |
|---|---|---|
| Restriction Enzymes | HaeIII, EcoRI [46] | DNA fragmentation; HaeIII shows higher precision for QX200 [46] |
| dPCR Master Mixes | Supermix for Probes (no dUTP) [3] | Critical for reaction efficiency and accuracy; performance varies by platform [3] |
| DNA Purification Kits | RSC PureFood GMO Kit [47] | Remove PCR inhibitors after restriction digestion [47] |
| Reference Assays | DCK, HBB, PMM1, RPS27A, RPPH1 [42] | Multiplex reference genes for copy number variation studies [42] |
| Digestion Buffers | Manufacturer-specific buffers [13] | Optimize restriction enzyme activity; compatibility with dPCR master mix is crucial [13] |
| Quantification Standards | gBlocks Gene Fragments [42] | Synthetic DNA fragments for standard curve generation and quality control [42] |
Experimental Protocol: Validating ddPCR Against PFGE for CNV Analysis
This section details the key experimental methodology from a 2025 study that established the high concordance of restriction enzyme-digital droplet PCR (ddPCR) with Pulsed-Field Gel Electrophoresis (PFGE) for Copy Number Variation (CNV) analysis [21].
- Study Design and Samples: The protocol utilized 40 genomic DNA samples from a clinical study cohort. These samples had previously been characterized for their copy number of the human alpha defensin 1–3 (DEFA1A3) gene using PFGE, which served as the reference method [21].
- Target Locus: The DEFA1A3 locus was used as a model. It is a multiallelic gene with a naturally varying copy number, generally ranging from 2 to 12 copies per diploid genome, making it ideal for evaluating CNV quantification techniques [21].
- ddPCR Procedure: The developed ddPCR protocol involved partitioning each PCR reaction into over 20,000 nanoliter-sized, water-in-oil droplets. This partitioning allows for absolute quantification of the target DNA molecule count using Poisson statistics. The protocol specifics, including primer and probe sequences, are detailed in the original study [21].
- Restriction Enzyme Digestion: As part of the sample preparation for ddPCR, genomic DNA was digested with restriction enzymes. This critical step reduces sample viscosity, prevents the uneven partitioning of large or linked DNA molecules, and ensures that individual gene copies are physically separated and can independently segregate into different droplets, leading to more accurate quantification [9].
- Data Analysis and Concordance Measurement: Copy numbers determined by ddPCR and a separate qPCR method were compared against the PFGE-derived results. Measurements were considered concordant if the ddPCR or qPCR result was within ±1 copy of the PFGE value. The relationship between methods was further evaluated using Spearman correlation and linear regression analysis [21].
Key Quantitative Findings: ddPCR Shows Superior Concordance with PFGE
The following table summarizes the core quantitative results from the validation study, demonstrating the performance of ddPCR compared to quantitative PCR (qPCR) when benchmarked against the PFGE gold standard [21].
| Metric | ddPCR vs. PFGE | qPCR vs. PFGE |
|---|---|---|
| Overall Concordance | 95% (38/40 samples) | 60% (24/40 samples) |
| Statistical Correlation (Spearman's r) | r = 0.90 (p < 0.0001) | r = 0.57 (p < 0.0001) |
| Average Difference from PFGE | 5% | 22% |
| Median of Differences | 0 (IQR [0, 0]) | -1.0 (IQR [-2, 1]) |
| Linear Regression Slope (vs. PFGE) | Y = 0.9953X | Y = 0.8889X |
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of a restriction enzyme-ddPCR workflow for CNV analysis requires careful selection of reagents. The table below lists key materials and their specific functions in the experimental process.
| Item | Function / Rationale |
|---|---|
| High-Quality Genomic DNA | Template for ddPCR; purity is critical as contaminants can inhibit PCR and interfere with fluorescence detection [9]. |
| Restriction Enzymes (e.g., HaeIII) | Fragments large DNA molecules to ensure even partitioning and physically separates tandemly-linked gene copies for accurate counting [4] [9]. |
| Sequence-Specific Hydrolysis Probes (TaqMan) | Provides sequence-specific detection in ddPCR, minimizing false positives from nonspecific amplification compared to DNA-binding dyes [9]. |
| ddPCR Supermix | A specialized PCR mix containing DNA polymerase, dNTPs, and buffer, optimized for the partitioning process and fluorescence signal generation in a droplet system. |
| Droplet Generator and Droplet Reader | Core instrumentation for creating thousands of nanoliter droplets and subsequently reading the fluorescence in each droplet to determine positives and negatives [21]. |
Troubleshooting Guide: FAQs for Restriction Enzyme-ddPCR
Q1: My ddPCR results show low precision and high coefficient of variation (CV). What could be the cause? A: Low precision can often be linked to the choice of restriction enzyme. A recent 2025 study found that using the HaeIII restriction enzyme significantly improved precision compared to EcoRI, especially in the QX200 ddPCR system. For the QIAcuity One system, the enzyme choice had less impact, but HaeIII still provided excellent results. Ensure your enzyme does not cut within your target amplicon sequence [4] [9].
Q2: Why is restriction enzyme digestion recommended for ddPCR sample preparation? A: Restriction digestion is crucial for several reasons [9]:
- Reduces Viscosity: Allows for accurate pipetting and partitioning of higher DNA concentrations.
- Separates Linked Copies: In tandemly repeated genes or arrays, digestion ensures that multiple copies are not trapped in a single droplet and counted as one, which would lead to under-quantification.
- Improves Partitioning: Large DNA molecules (>30 kb) can partition unevenly. Digestion creates smaller, more uniformly distributed fragments for accurate quantification.
Q3: My copy number values are consistently over-quantified. What should I check? A: Over-quantification is frequently associated with uneven partitioning of large DNA templates. Implementing a restriction enzyme digestion step is the primary solution to this problem, as it fragments the DNA for even distribution [9]. Additionally, verify that the average number of target copies per droplet is within the ideal range of 0.5 to 3 to avoid saturation, which can also affect accurate counting [9].
Q4: How does ddPCR performance compare to other common CNV detection methods? A: As validated against PFGE, ddPCR provides a unique combination of high accuracy, precision, and throughput [21] [49].
- vs. PFGE: ddPCR offers 95% concordance but is higher-throughput, less labor-intensive, and requires less specialized equipment [21].
- vs. qPCR: ddPCR demonstrates superior accuracy and precision, particularly at higher copy numbers, because it uses absolute quantification instead of relative Cq values, which can compound errors [21].
- vs. NGS: ddPCR is a low-cost, targeted method ideal for validating specific CNVs, whereas NGS can screen the entire genome but is more expensive and computationally intensive [21] [49].
Workflow Diagram: Restriction Enzyme-ddPCR for CNV Analysis
The diagram below illustrates the integrated experimental workflow, highlighting the critical role of the restriction enzyme digestion step.
Mechanism Diagram: How Restriction Enzymes Enhance Precision
This diagram details the mechanism by which restriction enzyme digestion improves the accuracy of copy number quantification in ddPCR.
FAQs: Restriction Enzymes and dPCR Precision
How does the choice of restriction enzyme affect dPCR precision? Research indicates that the choice of restriction enzyme can significantly impact the precision of your digital PCR results. A 2025 study comparing the QX200 ddPCR and QIAcuity One dPCR platforms found a general tendency for higher precision when using the HaeIII restriction enzyme instead of EcoRI. This effect was particularly pronounced for the QX200 system, where precision was greatly increased, with all coefficient of variation (CV) values falling below 5% when HaeIII was used [29].
When should I use a restriction enzyme in my dPCR assay? Incorporating a restriction digestion step prior to your dPCR run is recommended in several specific scenarios to ensure uniform template distribution, which is crucial for accurate quantification [9]:
- Linked or tandem gene copies: To physically separate gene copies so they can independently segregate into partitions.
- Highly viscous solutions: To reduce viscosity, allowing for more accurate pipetting and measurement, especially with high DNA concentrations.
- Supercoiled plasmids: To linearize the DNA, improving primer/probe binding efficiency.
- Large DNA molecules (>30 kb): To fragment large templates for more even partitioning and to prevent over-quantification.
Are dPCR systems robust to other experimental factors? Yes, multifactorial validation studies confirm that dPCR systems are highly robust. A systematic validation of the Bio-Rad QX200 Droplet dPCR system demonstrated that most experimental factors, including the operator, the primer/probe system, and the addition of restriction enzymes, have no relevant effect on the accuracy of DNA copy number quantification. This underscores the system's reliability across varying laboratory conditions [50].
Troubleshooting Guide: Improving dPCR Assay Performance
| Problem Area | Potential Cause | Recommended Solution |
|---|---|---|
| Sample Integrity | Linked gene copies or high molecular weight DNA causing uneven partitioning. | Use restriction enzymes (e.g., HaeIII) to fragment DNA. Ensure the enzyme does not cut within the amplicon sequence [29] [9]. |
| Sample Purity | Presence of PCR inhibitors (e.g., salts, humic acids, phenol). | Re-purify DNA using dedicated kits. dPCR is less prone to inhibition than qPCR, but high purity is still required for optimal fluorescence detection [9]. |
| Assay Precision | Suboptimal reaction conditions leading to high variation ("rain"). | Optimize annealing/extension temperature and oligonucleotide concentrations. Higher primer (0.5–0.9 µM) and probe (0.25 µM) concentrations can improve fluorescence amplitude and cluster separation [9] [51]. |
| Quantification Accuracy | Incorrect calculation of DNA input or copy number. | Calculate copy number from mass input using the genome size. For a single-copy gene, use the formula: Genome size (bp) x 1.096 x 10^–21 g/bp. Ensure the average copy per partition is between 0.5 and 3 for optimal quantification [9] [16]. |
| Signal Detection | Poorly separated positive and negative clusters. | Check for incompatible fluorophore/quencher combinations that create background noise. For probe-based assays, ensure thorough cleavage. For dye-based assays, achieve high PCR specificity to avoid signal from nonspecific products [9]. |
Experimental Protocol: Evaluating Restriction Enzymes in dPCR
The following methodology is adapted from a 2025 study that directly investigated the effect of restriction enzymes on dPCR precision [29].
1. Experimental Design:
- Platforms: Bio-Rad QX200 Droplet Digital PCR (ddPCR) and QIAGEN QIAcuity One Nanoplate digital PCR (ndPCR).
- Template DNA: DNA extracted from varying cell numbers of the ciliate Paramecium tetraurelia and synthetic oligonucleotides.
- Key Variable: Treatment with different restriction enzymes (e.g., EcoRI vs. HaeIII).
2. Materials and Reagents:
- dPCR Systems: QX200 ddPCR system or QIAcuity One system.
- Restriction Enzymes: EcoRI and HaeIII.
- Master Mix: Use the master mix recommended for the platform. Note that the choice of master mix can be a critical factor for accuracy [50].
- Primers/Probes: Assay specific to the target gene.
3. Step-by-Step Procedure:
- Step 1: DNA Digestion. Treat aliquots of the sample DNA with the different restriction enzymes according to the manufacturer's protocols.
- Step 2: Reaction Setup. Prepare dPCR reaction mixes containing:
- dPCR master mix
- Optimized concentrations of primers and probes
- A fixed volume of undigested or digested DNA
- Step 3: Partitioning and Amplification. Load reactions onto the dPCR instrument for partitioning, followed by endpoint PCR amplification using optimized thermal cycling conditions.
- Step 4: Imaging and Analysis. The instrument images the partitions and software counts the positive and negative reactions to calculate the absolute copy concentration.
4. Data Analysis:
- Calculate the Coefficient of Variation (CV) for replicate measurements for each restriction enzyme condition.
- Compare the CV values between enzymes (e.g., EcoRI vs. HaeIII) and across platforms to assess which combination yields the highest precision.
The workflow below summarizes the key steps in this experimental protocol:
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function in dPCR | Application Note |
|---|---|---|
| Restriction Enzymes (e.g., HaeIII) | Fragments genomic DNA to ensure random partitioning of linked gene copies and reduce viscosity. | Critical for improving precision, especially with complex templates. Must not cut within the amplicon [29] [9]. |
| dPCR Master Mix | Provides optimized buffer, polymerase, and dNTPs for efficient amplification within partitions. | A critical performance factor. Validation studies show the choice of master mix can directly affect accuracy [50]. |
| Hydrolysis Probes (TaqMan) | Provide sequence-specific detection through fluorophore/quencher separation during amplification. | Reduce background vs. intercalating dyes. Avoid reporter/quencher emission overlap to prevent noise [9]. |
| QIAcuity Nanoplate / Bio-Rad QX200 Cartridge | Creates thousands of individual reaction chambers (partitions) for single-molecule PCR. | The core of dPCR technology. Platform choice (nanoplate vs. droplet) does not preclude high precision when methods are optimized [29] [47]. |
In the field of molecular diagnostics and genetic research, accurately quantifying gene copy numbers is essential for understanding disease mechanisms, organismal abundance, and genetic diversity. For years, quantitative PCR (qPCR) has been the standard method for this task. However, when dealing with samples containing very high gene copy numbers, such as those from microorganisms with highly amplified genomes or in cancer research with gene amplifications, qPCR faces significant challenges in precision and accuracy.
Digital PCR (dPCR), a technique that partitions a sample into thousands of individual reactions for absolute nucleic acid quantification, presents a powerful alternative. Recent research indicates that the combination of dPCR with specific restriction enzymes significantly enhances its performance, enabling it to outperform qPCR in the analysis of high-copy-number targets. This technical article explores the evidence behind this advantage, providing troubleshooting guidance and detailed protocols for researchers aiming to implement this method.
### Fundamental Principles
Quantitative PCR (qPCR) estimates the initial amount of a DNA target by monitoring the amplification in real-time during the exponential phase. The cycle threshold (Ct) at which the fluorescence signal crosses a defined threshold is compared to a standard curve to determine the relative concentration [52] [53]. This reliance on a standard curve and amplification efficiency can introduce variability.
Digital PCR (dPCR), in contrast, provides absolute quantification without the need for a standard curve. The sample is partitioned into tens of thousands of nanoreactions. After an end-point PCR amplification, each partition is analyzed as positive or negative for the target. The absolute concentration of the target, in copies per microliter, is then calculated directly using Poisson statistics [4] [15] [53].
### The Critical Role of Restriction Enzymes in dPCR
The precision of dPCR, especially for complex genomic regions or organisms with high inherent copy number variation, can be substantially improved by using restriction enzymes during sample preparation [4]. These enzymes digest the DNA, breaking up tangled long strands and inaccessible secondary structures. This process ensures a more random distribution of DNA molecules during the partitioning step, which is critical for accurate Poisson correction and final copy number calculation.
Diagram 1: Core dPCR workflow with integrated restriction enzyme digestion.
## Experimental Evidence: A Case Study in Protist Research
A 2025 study directly compared the precision of two dPCR platforms (droplet-based QX200 and nanoplate-based QIAcuity One) and evaluated the impact of two restriction enzymes (EcoRI and HaeIII) on copy number quantification using the ciliate Paramecium tetraurelia, an organism known for its high gene copy number variability [4].
### Key Experimental Protocol
- DNA Source: Synthetic oligonucleotides and DNA extracted from varying cell numbers (0, 1, 5, 10, 50, 100) of P. tetraurelia.
- Restriction Enzymes Tested: EcoRI and HaeIII.
- dPCR Platforms: QX200 Droplet Digital PCR (Bio-Rad) and QIAcuity One nanoplate-based dPCR (QIAGEN).
- Key Metrics: Limit of Detection (LOD), Limit of Quantification (LOQ), accuracy (deviation from expected value), and precision (Coefficient of Variation, %CV).
The following table summarizes the key performance metrics from the study, highlighting the critical role of restriction enzyme selection.
Table 1: Comparative Performance Metrics of dPCR Platforms [4]
| Metric | QIAcuity One (ndPCR) | QX200 (ddPCR) | Notes |
|---|---|---|---|
| Limit of Detection (LOD) | 0.39 copies/µL input | 0.17 copies/µL input | Sensitivity for detecting the target. |
| Limit of Quantification (LOQ) | 1.35 copies/µL input | 4.26 copies/µL input | Minimum concentration for reliable quantification. |
| Precision with EcoRI (Avg. %CV) | ~0.6% - 27.7% | ~2.5% - 62.1% | High variability, especially at medium cell counts with ddPCR. |
| Precision with HaeIII (Avg. %CV) | ~1.6% - 14.6% | < 5% (all cell counts) | HaeIII drastically improved precision for ddPCR. |
The most significant finding was the dramatic effect of the restriction enzyme on assay precision. While the choice of enzyme had a minor effect on the QIAcuity One platform, it was critical for the QX200 system. Using HaeIII instead of EcoRI reduced the Coefficient of Variation (CV) for the QX200 to below 5% for all tested cell numbers, indicating superior precision and reproducibility [4].
## Troubleshooting Guide & FAQs
Q1: Why does dPCR outperform qPCR for high copy number quantification?
The core advantage lies in the method of quantification. qPCR relies on a standard curve and the assumption of consistent amplification efficiency, which can break down with complex, high-copy-number templates, leading to high variability and inaccurate results [5] [52]. dPCR uses absolute counting and Poisson statistics, which are less affected by amplification efficiency variations. The partitioning step in dPCR effectively dilutes the sample, reducing the impact of PCR inhibitors and making the quantification more robust [54] [27].
Q2: Why is a restriction enzyme necessary in my dPCR assay, and how do I choose one?
Restriction enzymes digest long DNA strands, promoting a random distribution of target molecules during partitioning. This is vital for accurate Poisson correction [4].
- Troubleshooting Tip: If you observe high CV values or inconsistent results between replicates, the DNA may not be digesting efficiently.
- Solution: Test different restriction enzymes. The optimal enzyme is one that cuts efficiently in your target region but does not cut within the amplicon itself. As shown in the case study, enzyme choice can be a decisive factor for precision [4].
Q3: My dPCR results show high variation between replicates. What could be the cause?
High variation can stem from several sources. Follow this diagnostic workflow to identify the issue.
Diagram 2: Troubleshooting high variation in dPCR results.
Q4: When should I choose qPCR over dPCR for copy number analysis?
The choice depends on your project's needs.
- Choose qPCR for: High-throughput, cost-sensitive projects where relative quantification is sufficient, and the target is present in moderate to high abundance [52] [53].
- Choose dPCR for: Applications requiring absolute quantification, high precision, detection of small fold-changes (e.g., from 5 to 6 copies), analysis of low-abundance targets, or work with difficult samples that may contain inhibitors [5] [27] [53].
## The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for Restriction Enzyme-dPCR Experiments
| Reagent / Material | Function | Example from Literature |
|---|---|---|
| Restriction Enzymes | Digests genomic DNA to ensure random distribution of target molecules for precise Poisson-based quantification. | HaeIII, EcoRI [4] |
| Digital PCR System | Partitions the sample and performs endpoint PCR and fluorescence reading. | QIAcuity One (QIAGEN), QX200 (Bio-Rad) [4] [54] |
| Fluorescent Probe/Primer Assays | Provides target-specific amplification and detection in partitioned reactions. | TaqMan Probe Assays [55] |
| High-Quality DNA Extraction Kit | Ishes pure, intact genomic DNA suitable for restriction digestion and PCR amplification. | Kits from KingFisher, STARlet platforms [54] |
Conclusion
The integration of restriction enzymes is a critical, evidence-based strategy for maximizing the precision of digital PCR, particularly for complex genomic DNA targets. By enzymatically fragmenting DNA to enhance target accessibility, researchers can overcome a fundamental limitation of dPCR, leading to significantly improved data reproducibility and accuracy across platforms like the Bio-Rad QX200 and QIAGEN QIAcuity. The choice of enzyme, exemplified by the superior performance of HaeIII over EcoRI in certain systems, is a key determinist of success. As dPCR cements its role in clinical diagnostics—from liquid biopsy to CNV analysis and epigenetics—the optimized use of restriction enzymes will be paramount for developing robust, high-throughput assays that deliver reliable, actionable data for biomedical research and therapeutic development.
References
- 1. dPCR: A Technology Review - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC5948698/]
- 2. Medical diagnostic value of digital PCR (dPCR) [https://www.sciencedirect.com/science/article/pii/S2667099223000221]
- 3. In-house validation of a droplet digital PCR system using a ... [https://www.sciencedirect.com/science/article/pii/S0003267025005665]
- 4. Comparing the precision of two digital PCR applications for ... [https://www.nature.com/articles/s41598-025-13143-8]
- 5. Digital droplet PCR is an accurate and precise method to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12546813/]
- 6. PCR Troubleshooting Guide | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/pcr-education/pcr-reagents-enzymes/pcr-troubleshooting.html]
- 7. Troubleshooting your PCR [https://www.takarabio.com/learning-centers/pcr/faq/troubleshooting?srsltid=AfmBOorVxyxaI8tknhS8DOLmjeF6TU6p_PnRzXurJxTFLvFd9FozU-aD]
- 8. PCR Troubleshooting Guide [https://www.neb.com/en-us/tools-and-resources/troubleshooting-guides/pcr-troubleshooting-guide?srsltid=AfmBOoobhHBq3WBPIkpblkBGHLzjQwLWc_lIq4VOfVAFiIKq1CMOdLo9]
- 9. dPCR assay development | Digital PCR troubleshooting [https://www.qiagen.com/us/applications/digital-pcr/beginners/dpcr-guide/setup-and-troubleshooting]
- 10. Restriction Enzymes for Droplet Digital PCR (ddPCR) [https://www.neb.com/en-us/tools-and-resources/usage-guidelines/restriction-enzymes-for-droplet-digital-pcr?srsltid=AfmBOorU4dSuQRBuH7ijbxjFhrLVVnAbMr_9B6PICBKXZjxL9yJu5lLq]
- 11. Restriction Enzymes in Droplet Digital PCR [https://www.neb.com/en-us/tools-and-resources/video-library/nebtv-episode-15-restriction-enzymes-in-droplet-digital-pcr?srsltid=AfmBOorED2ua9Q3wO7hhuNRn080Cj03q3w4yvtDJOFyJ05d1-AfVdw8C]
- 12. Evaluating Digital PCR for the quantification of human ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5108050/]
- 13. Restriction enzyme troubleshooting guide [https://www.thermofisher.com/us/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/molecular-cloning/restriction-enzymes/restriction-enzyme-troubleshooting-guide.html]
- 14. Fix Failed DNA Digests: Restriction Enzyme Troubleshooting [https://go.zageno.com/blog/restriction-enzyme-digestion-troubleshooting]
- 15. Digital PCR: from early developments to its future application ... [https://pubs.rsc.org/en/content/articlehtml/2025/lc/d5lc00055f]
- 16. Digital PCR Support—Troubleshooting [https://www.thermofisher.com/us/en/home/technical-resources/technical-reference-library/real-time-digital-PCR-applications-support-center/digital-pcr-support/digital-pcr-support-troubleshooting.html]
- 17. Digital PCR outperforms quantitative real-time PCR for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12288185/]
- 18. Restriction Enzymes in Droplet Digital PCR [https://www.neb.com/en/tools-and-resources/video-library/nebtv-episode-15-restriction-enzymes-in-droplet-digital-pcr?srsltid=AfmBOorEu-Lgcf2FPf-1xIuZuV0RPZ9Ua216FEVnjm2XmQgy_WAiw_vN]
- 19. Troubleshooting Common Issues with Restriction Digestion ... [https://www.thermofisher.com/us/en/home/brands/thermo-scientific/molecular-biology/molecular-biology-learning-center/molecular-biology-resource-library/spotlight-articles/7-common-issues-with-restriction-digestion-reactions-and-how-to-avoid-them.html]
- 20. Restriction Digestion Troubleshooting Guide [https://www.genscript.com/restriction-digestion-troubleshooting-guide.html]
- 21. Digital droplet PCR is an accurate and precise method to ... [https://www.nature.com/articles/s41598-025-20944-4]
- 22. Digital PCR Market Size, Forecast Report [https://www.mordorintelligence.com/industry-reports/digital-pcr-market]
- 23. Restriction Enzyme Digestion Key Considerations [https://www.thermofisher.com/us/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/molecular-cloning/restriction-enzymes/restriction-enzyme-key-considerations.html]
- 24. qPCR Troubleshooting Guide [https://azurebiosystems.com/qpcr-resources/troubleshooting/]
- 25. Restriction Enzymes for Droplet Digital PCR (ddPCR) [https://www.neb.com/en-us/tools-and-resources/usage-guidelines/restriction-enzymes-for-droplet-digital-pcr?srsltid=AfmBOorjdiHrAsxIGj6iChz3hnbA4nNYhXK5jZxwWwGpkCEzka2Si2GQ]
- 26. PCR Troubleshooting Guide [https://www.neb.com/en-us/tools-and-resources/troubleshooting-guides/pcr-troubleshooting-guide?srsltid=AfmBOortT3cZ-fSJeZ57yfn_kKBDkIVqrbhqDsOUtwqE9BTyFWLPxfG9]
- 27. Digital PCR Applications: Copy Number Variation [https://www.thermofisher.com/us/en/home/life-science/pcr/digital-pcr/education/basic-research/copy-number-variation.html]
- 28. How to Troubleshoot Sequencing Preparation Errors (NGS ... [https://www.cd-genomics.com/resource-sequencing-preparation-troubleshooting.html]
- 29. Comparing the precision of two digital PCR applications for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12297429/]
- 30. Absolute quantification reveals the stable transmission of a ... [https://bmcgenomics.biomedcentral.com/articles/10.1186/s12864-016-2619-0]
- 31. Copy Number Variation (CNV) Support [https://www.thermofisher.com/us/en/home/technical-resources/technical-reference-library/real-time-digital-PCR-applications-support-center/copy-number-variation-support.html]
- 32. CNV Workflow [https://support-docs.illumina.com/SW/DRAGEN_v39/Content/SW/DRAGEN/CNVWorkflow.htm]
- 33. Digital PCR: from early developments to its future application ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12278255/]
- 34. Methylation-sensitive restriction enzyme-droplet digital ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10998716/]
- 35. Methylation‑sensitive restriction enzyme‑droplet digital ... [https://www.spandidos-publications.com/10.3892/ijmm.2024.5366]
- 36. Comparative Analysis of Two Digital PCR Platforms for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12361875/]
- 37. A validated restriction enzyme ddPCR cg05575921 (AHRR ... [https://clinicalepigeneticsjournal.biomedcentral.com/articles/10.1186/s13148-024-01659-1]
- 38. Using qPCR and ddPCR to study biodistribution of cell ... [https://www.sciencedirect.com/science/article/pii/S1465324924008648]
- 39. HaeIII [https://en.wikipedia.org/wiki/HaeIII]
- 40. The Restriction Endonuclease Alu I Induces Chromosomal ... [https://pubmed.ncbi.nlm.nih.gov/3023993/]
- 41. Csp6I (CviQI) (10 U/μL) [https://www.thermofisher.com/order/catalog/product/ER0211]
- 42. Multiplexed Digital PCR Reference Gene Measurement for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12523864/]
- 43. PCR tips and tricks (incl. master mix calculations) | INTEGRA [https://www.integra-biosciences.com/united-states/en/blog/article/pcr-tips]
- 44. Troubleshooting Common Issues with Restriction Digestion ... [https://www.thermofisher.com/sa/en/home/brands/thermo-scientific/molecular-biology/molecular-biology-learning-center/molecular-biology-resource-library/spotlight-articles/7-common-issues-with-restriction-digestion-reactions-and-how-to-avoid-them.html]
- 45. Restriction enzyme troubleshooting guide [https://www.thermofisher.com/es/en/home/life-science/cloning/cloning-learning-center/invitrogen-school-of-molecular-biology/molecular-cloning/restriction-enzymes/restriction-enzyme-troubleshooting-guide.html]
- 46. Comparing the precision of two digital PCR applications for ... [https://pubmed.ncbi.nlm.nih.gov/40715338/]
- 47. Comparison of two digital PCR platforms for quantification ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12622311/]
- 48. Development and validation of a droplet digital PCR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11532291/]
- 49. Copy Number Variation: Methods and Clinical Applications [https://www.mdpi.com/2076-3417/11/2/819]
- 50. In-house validation of a droplet digital PCR system using ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267025005665]
- 51. Optimization of digital droplet polymerase chain reaction for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4827695/]
- 52. Quantitative or digital PCR? A comparative analysis for ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993624001584]
- 53. Digital PCR (dPCR) vs Real-Time PCR (qPCR) [https://firalismolecularprecision.com/2025/04/09/dpcr-qpcr-differences/]
- 54. Comparative Performance of Digital PCR and Real-Time RT ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12474457/]
- 55. Comparison of real-time quantitative PCR and two digital ... [https://www.sciencedirect.com/science/article/pii/S0022175924000139]